��Ŀ����
20��ijУ�о���ѧϰС��Ϊ�˲ⶨ���ؿ�ɽʯ��ʯ��̼��Ƶ�����������ȡ����һЩ��ʯ��Ʒ����ȡϡ����200g��ƽ���ֳ�4�ݽ���ʵ�飬������£�| ʵ���� | 1 | 2 | 3 | 4 |
| ������Ʒ������/g | 5 | 10 | 15 | 20 |
| ����CO2������/g | 1.76 | 3.52 | 4.4 | m |
��1���ϱ���m����ֵ��4.4��
��2����Ӧ�п�ʯ��ʣ����ǵ�3��4��ʵ�飻
��3����������ʯ��ʯ��̼��Ƶ�����������
��4������ϡ���������ʵ�����������
���� ȡϡ����200 g��ƽ���ֳ�4�ݽ���ʵ�飬ÿ��ʵ��ʹ��ϡ����50g��
����ʵ�������ݱ���5gʯ��ʯ��ȫ��Ӧ�������ɶ�����̼1.76g����15gʯ��ʯ��ȫ��Ӧ���ɶ�����̼����=1.76g��3=5.28g����ʵ���н��ų�4.4g������̼��˵����ʱʯ��ʯû����ȫ��Ӧ��û����ȫ��Ӧ������ϡ����㣻
���ݷ�Ӧ����ط�������ݿ��Լ���̼��Ƶ��������������Ȼ������������һ�����Լ���ʯ��ʯ����̼��Ƶ�����������ϡ���������ʵ�����������
��� �⣺��1����ʯ��ʯ����Ϊ15gʱ������50gϡ�����������ʯ��ʯû��ȫ��Ӧ�����ԣ�ʯ��ʯ���ӵ�20gʱ��50gϡ������Ȼ�����㣬���Դ�ʱ����������̼��������ȻΪ4.4g��
�ʴ�4.4��
��2��������ʵ��50gϡ����������ʯ��ʯû��ȫ��Ӧ�����Ĵ�ʵ��ʯ��ʯ���ӵ�20gʱ��50gϡ������Ȼ�������ʯ��ʯû����ȫ��Ӧ��
�ʴ������ģ�
��3����10gʯ��ʯ��̼��Ƶ�����Ϊx
CaCO3+2HCl=CaCl2+H2O+CO2��
100 44
x 3.52g
$\frac{100}{x}=\frac{44}{3.25g}$
��֮�� x=8g
ʯ��ʯ����̼��Ƶ���������=$\frac{8g}{10g}$��100%=80%��
��4���Ե��������ݼ��㣺
��50gϡ�������Ȼ��������Ϊy��
CaCO3+2HCl=CaCl2+H2O+CO2��
73 44
y 4.4g
$\frac{73}{y}=\frac{44}{4.4g}$
��֮�� y=7.3g
ϡ���������ʵ���������=$\frac{7.3g}{50g}$��100%=14.6%��
�𰸣�
��1��4.4��
��2��3��4��
��3������ʯ��ʯ��̼��Ƶ���������Ϊ80%��
��4��ϡ���������ʵ���������Ϊ14.6%��
���� ǰ����ʵ��̼�����ȫ��Ӧ���ɸ��ݴ�ʱ�ķ�Ӧ����ʯ��ʯ��̼��Ƶ����������������ʵ����������ȫ��Ӧ�����ݴ�ʱ�ķ�Ӧ����ϡ�������������������

| A�� | ͨ��ʳ��ˮ | B�� | ͨ������������Һ | ||
| C�� | ��ȼ������� | D�� | ͨ�������ʯ��ˮ |
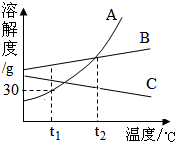 A��B��C���ֲ����ᾧˮ�Ĺ������ʵ��ܽ��������ͼ������˵����ȷ���ǣ�������
A��B��C���ֲ����ᾧˮ�Ĺ������ʵ��ܽ��������ͼ������˵����ȷ���ǣ�������| A�� | t1��ʱ��A�ı�����Һ100g�к�������30g | |
| B�� | ��t2�棬A��B������Һ�����ʵ�����������ͬ | |
| C�� | Ҫ��B��Һ�еõ�B��ͨ���ɲ��ý���ʹ��ᾧ�ķ��� | |
| D�� | ��t2��ʱA��B��C�������ʵı�����Һ������t1��ʱ��A��Һ�����ʵ�����������С |
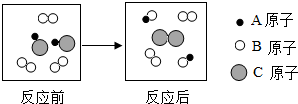
| A�� | �÷�Ӧ������û�е������� | B�� | �÷�Ӧ�����з�������û�б仯 | ||
| C�� | �÷�Ӧ������ԭ��������˱仯 | D�� | �÷�Ӧ�����û���Ӧ |
| A�� | 0.8g | B�� | 1.2g | C�� | 2.4g | D�� | 3.6g |
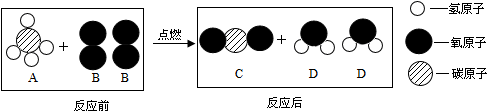
| A�� | ��ȼAǰ��Ҫ�ȼ��鴿�� | B�� | ͼʾ�й���4�ֻ�������� | ||
| C�� | ͼʾ����һ���л��� | D�� | ˵����ѧ��Ӧ�з��ӿɷ� |
| A�� | ʯī�ͽ��ʯ�������ʲ�ͬ--̼ԭ�ӽṹ��ͬ | |
| B�� | һ����̼�Ͷ�����̼�Ļ�ѧ���ʲ�ͬ--���ӹ��ɲ�ͬ | |
| C�� | ��CO2����NaOH��Ca��OH��2��Һ--CO2ֻ��Ca��OH��2��Ӧ | |
| D�� | �÷�̪��Һ���ܼ���ϡ�����ϡ����--��̪��Һ�������ᶼ���ɫ |
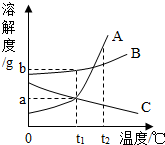 ��ͼ��ʾΪA��B��C���ֹ������ʣ������ᾧˮ�����ܽ�����ߣ������й�˵����ȷ����B
��ͼ��ʾΪA��B��C���ֹ������ʣ������ᾧˮ�����ܽ�����ߣ������й�˵����ȷ����B