��Ŀ����
Ϊ�˲ⶨʯ��ʯ�к�CaCO3 ����������,ȡʯ��ʯ12.5g ����ʢ��100g ϡ�� ����ձ���,����ǡ����ȫ��Ӧ���������ʲ������ᷴӦ��Ҳ���ܽ⣩���ձ������ʵ�������Ϊ108.1g ��
����ձ���,����ǡ����ȫ��Ӧ���������ʲ������ᷴӦ��Ҳ���ܽ⣩���ձ������ʵ�������Ϊ108.1g ��
�� ��Ӧ�Ļ�ѧ����ʽΪ________________________________��
�� ���ɶ�����̼����________g��
�� 12.5gʯ��ʯ��CaCO3 ________g��ʯ��ʯ��CaCO3 ����������Ϊ________��
�� 100g ϡ������������________����������________g���ܼ���______ __����������________g��
__����������________g��
�� ��ȫ��Ӧ���ձ��ڻ��в������ʣ��ձ���������Һ������Ϊ________��������Һ������Ϊ
________g���������ʵ�����Ϊ________g���ܼ�������Ϊ________g��
CaCO3 + 2HCl =CaCl2 + H2O + CO2����4.4g��10g ��80%���Ȼ��⡢7.3g��ˮ��92.7g���Ȼ�����Һ��105.6g�� 11.1g��94.5g��
11.1g��94.5g��

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ

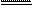 2Cl2 + 2Ca(OH)2 Ca(ClO)2 + X + 2H2O������X�Ļ�ѧʽΪ��������
2Cl2 + 2Ca(OH)2 Ca(ClO)2 + X + 2H2O������X�Ļ�ѧʽΪ�������� ��������ԭ�ԡ��������ԡ�����
��������ԭ�ԡ��������ԡ�����