��Ŀ����
1����1���û�ѧ�����ʾ���ٽ�Ԫ��Au��
��2��ˮ����2H2O��
��ijЩʳƷ��װ����һС����������ɷ�Ϊ��ʯ�ң���������ˮ�Ļ�ѧ����ʽΪCaO+H2O=Ca��OH��2��
��2����ѧ������ϢϢ��أ�����ѧ���Ļ�ѧ֪ʶ����������⣮
�ٲ����ϵ�ʳ���У��������������⡢�����㡢������ΪʹӪ�����⣬����ΪӦ�����ӵ�ʳ�����߲��ࣨ��ˮ���ࣩ��д��һ�֣���
���ڱ����з����û���̿�Ƴɵġ��������������Ա��������ζ�����������ã�
�ۼ�ͥ�����õ������ֱ������ȹ��� ����ȹ��ԡ��������ԡ����������ģ�
���� ���⿼�黯ѧ��������弰��д������ؼ��Ƿ��廯ѧ����������Ķ����Ƿ��ӡ�ԭ�ӡ����ӻ��ǻ��ϼۣ������ڻ�ѧ����ǰ������λ�ü����ʵ��ļ������������ر��������壬���ܸ������ʻ�ѧʽ����д������ȷ��д���ʵĻ�ѧʽ����������ȷ�Ľ�������Ŀ����Ϥʳ���е�Ӫ���ء�����̿�����������Լ����ϵ�����ɣ�
��� �⣺��1���٢ٽ�Ԫ�صķ���ΪAu�����Au��
��2��ˮ���Ӿ�����ˮ�Ļ�ѧʽ��ǰ���������2�����2H2O��
������������ˮ��Ӧ�����������ƣ����CaO+H2O=Ca��OH��2��
�ٶ��������������ʡ�ˮ�����Σ������⸻�������ʺ���֬�������㸻�������ʣ������������࣬ȱ�ٵ���ά���������ʣ�����Ҫ�����߲˻���ˮ��������߲��ࣨ��ˮ���ࣩ��
�ڻ���̿���������ԣ������������е���ζ�����������
�ۼ�ͥ�����õ������ֱ������ȹ����������ɵģ�����ȹ��ԣ�
���� ������Ҫ����ѧ���Ի�ѧ�������д�����������Լ���ѧ�������֪ʶ����Ŀ��ƼȰ����Ի�ѧ����������˽⣬�ֿ�����ѧ���Ի�ѧ���ŵ���д������ȫ�棬ע�ػ�������Ŀ�ѶȽ��ף�

��ϰ��ϵ�д�
�����Ŀ
12����ͼ��10������Ͳ�ľֲ�������ͼʾ��Һ�������ǣ�������
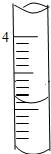

| A�� | 3.1ml | B�� | 5.1ml | C�� | 4.1ml | D�� | 2.2ml |
16�����л�ѧ��Ӧ�Ļ������Ͱ��������Ϸ�Ӧ���ֽⷴӦ���û���Ӧ���ֽⷴӦ�����з�Ӧ�в����������ֻ������͵��ǣ�������
| A�� | CO2+H2O�TH2CO3 | B�� | CH4+2O2$\frac{\underline{\;��ȼ\;}}{\;}$CO2+2H2O | ||
| C�� | 2H2O $\stackrel{ͨ��}{��}$2H2��+O2�� | D�� | FeS+H2SO4�TFeSO4+H2S�� |
 ijУ��ȤС��ӷϾ����Ͻ���һ����Ƭ����������ƽ��������Ϊ27g��Ȼ��������21.9%��ϡ�����У��������������������������������ͼ���������Ƭ�г����溬���������⣬�������ʲ����ᷴӦ�Ҳ�����Ԫ�أ�����ش�
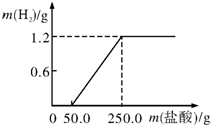
ijУ��ȤС��ӷϾ����Ͻ���һ����Ƭ����������ƽ��������Ϊ27g��Ȼ��������21.9%��ϡ�����У��������������������������������ͼ���������Ƭ�г����溬���������⣬�������ʲ����ᷴӦ�Ҳ�����Ԫ�أ�����ش�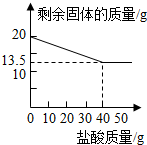 ij��ѧ��ȤС���ͬѧΪ�ⶨ�ٻƽ�ͭп�Ͻ����Ԫ�ص�������������ȡ20�˼ٻƽ������ձ��У���ijŨ�ȵ�ϡ����50�ˣ���5�μ��룬ÿ�γ�ַ�Ӧ��ȡ�����壬�����ˡ�����Ȳ�������أ�����ϡ����������ʣ����������Ĺ�ϵ��ͼ��ʾ��
ij��ѧ��ȤС���ͬѧΪ�ⶨ�ٻƽ�ͭп�Ͻ����Ԫ�ص�������������ȡ20�˼ٻƽ������ձ��У���ijŨ�ȵ�ϡ����50�ˣ���5�μ��룬ÿ�γ�ַ�Ӧ��ȡ�����壬�����ˡ�����Ȳ�������أ�����ϡ����������ʣ����������Ĺ�ϵ��ͼ��ʾ��