��Ŀ����
���������������еõ��㷺Ӧ�á�
��1���������Ƴ����������������˽������������õ� ��
��2������Ʒ����������� ��������ϡ��������⣬д���������Ļ�ѧ����ʽ ��
��3���ҹ����������������ʪ��ұ����ȡͭ�Ĺ��ң���������ͭ��Һ��Ӧ�Ļ�ѧ����ʽΪ ��
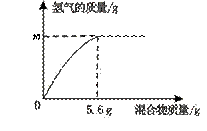 ��4���������ֽ���������ɵĻ�����100 gijϡ�����м���û�������������������������������ϵ��ͼ��ʾ��
��4���������ֽ���������ɵĻ�����100 gijϡ�����м���û�������������������������������ϵ��ͼ��ʾ��
����˵������ȷ��һ���� ��
a. �������ΪZn��Al����m������0.2 g
b. �������ΪZn��Cu����m=0.2 g
c. �������ΪFe��Al����ϡ������������
������һ������7.3%
d. �������ΪFe��Cu��m=0.1 g����������Fe����������Ϊ50%
���㣺
�������������ʼ���;�������Ļ�ѧ���ʣ�������ʴ�����������������Ļ�ѧ���ʣ���д��ѧ����ʽ�����ֱ���ʽ�����뷽��ʽ��.
ר�⣺
������������ϣ�
������
��1������������������õ���չ�ԣ�
��2��������ˮ����������ʱ�����⣻�������Ҫ�ɷ������������������ᷴӦ�����Ȼ�����ˮ���ݴ˽��
��3�����Ļ�Ա�ͭǿ����������ͭ��Ӧ��������������ͭ��д����Ӧ�Ļ�ѧ����ʽ���ɣ�
��4����Ϊ�������ԭ������������п��С��������ͬ����������������ȫ��Ӧʱ�����������������࣮
���
�⣺��1���������Ƴ����������������˽������������õ� ��չ�ԣ�
��2��������ˮ����������ʱ�����⣻�������Ҫ�ɷ������������������ᷴӦ�����Ȼ�����ˮ�����Fe2O3+6HCl�T2FeCl3+3H2O��
��3�����Ļ�Ա�ͭǿ����������ͭ��Ӧ��������������ͭ����Ӧ�Ļ�ѧ����ʽΪ��Fe+CuSO4�TFeSO4+Cu��
��4���������ȫ����������������Ϊ5.6 g������ݻ�ѧ��Ӧ�ķ���ʽ��Fe+2HCl=FeCl2+H2���ɼ������ʱ������������������0.2 g��ͬ���ɼ����5.6 gAl�����ᷴӦ������������������0.2 g��5.6gп�����ᷴӦ��������������С��0.2 g��ͭ�������Ӧ��
a���������ΪZn��Al��m���ܵ���0.2g����a��ȷ��
b���������ΪZn��Cu����m����0.2g����b����
c������ ����ΪFe��Alʱ������ͬ������������ϡ����������������������������5.6 g��ʱ
����ΪFe��Alʱ������ͬ������������ϡ����������������������������5.6 g��ʱ ����
���� �����������Ϊ7.3 g�����Ի����ΪFe��Alʱ�������������������7.3 g����������������ʽ��֪��������������������һ������7.3%����c��ȷ��
�����������Ϊ7.3 g�����Ի����ΪFe��Alʱ�������������������7.3 g����������������ʽ��֪��������������������һ������7.3%����c��ȷ��
d���������ΪFe��Cu��mΪ0.1g������0.1g������Ҫ����������2.8g����������������������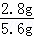 ��100%=50%����d��ȷ��
��100%=50%����d��ȷ��
�ʴ�Ϊ����1����չ�ԣ���2������������ˮͬʱ�Ӵ���Fe2O3+6HCl�T2FeCl3+3H2O����3��Fe+CuSO4�TFeSO4+Cu����4��acd
������
��ͬ�����Ľ������ᷴӦ���������������Ķ�������������ԭ�������йأ������ԭ������Խ��������������ԽС�����⿼����������������Լ�

�ִ���������������Ҫ���Ĵ������������л�У�ͨ����ѧ��Ӧ�ṩ�������ǣ�������
| �� |
A�����亽��ɻ� |
B��ˮ������ |
C��̫���ܹ��� |
D���������� |
��ѧ������������أ���������ѧ��ѧ֪ʶ��������жϣ�����˵����ȷ���ǣ�������
| �� | A | θ�����ɷ�������������Һ�����к� |
| �� | B | ���̶����彡��û��Ӱ�죬��Ϊȼ�ŵ����̲����������в����������к������� |
| �� | C | ��¥�������Ӧ�������Ŵ�ͨ�磬����ʪë����ס�ڱǣ����¿������������ǽ�������Ż����� |
| �� | D | ��������Ϊ������֯�ṩӪ����������������ҺΪ���˲���Ѫ�� |




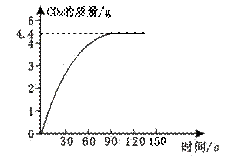 ijУ��ѧ��ȤС��ͬѧ���֣�����ʹ�õ���ˮ���ײ���һ��ˮ����ˮ������Ҫ�ɷ���̼��ƺ�������þ������Ϊ�˲ⶨˮ����̼��Ƶĺ�����ȡij��ϡ����100 g���뵽15.8 gˮ���У�����CO2������������ͼ��ʾ�����㣺
ijУ��ѧ��ȤС��ͬѧ���֣�����ʹ�õ���ˮ���ײ���һ��ˮ����ˮ������Ҫ�ɷ���̼��ƺ�������þ������Ϊ�˲ⶨˮ����̼��Ƶĺ�����ȡij��ϡ����100 g���뵽15.8 gˮ���У�����CO2������������ͼ��ʾ�����㣺