��Ŀ����
17�������ҹ��Ϸ����ʡ�����ܴ�Χ�����ɺ�����������ˮ��ȫ���ܹ�ע����1����ȥˮ�������Թ������ʵIJ����ǹ��ˣ�
��2������ˮ����Ӳˮ������ˮ�����õ������Ƿ���ˮ�������н�Ӳˮת��Ϊ��ˮ������У�
��3�����й���ˮ��˵����ȷ����B��
A��ˮ���л���ܸ�������
B������ˮ��Դ��Ҫ��Լ��ˮ����Ҫ��ֹˮ����Ⱦ
C��ˮ������ԭ�Ӻ���ԭ�ӹ��ɵ�
D��ˮ����Ԫ�غ����ܸߣ����ṩ���������������
���� ��1�������ǰѲ�����Һ��Ĺ�����Һ������һ�ַ������ݴ˽��з������
��2������Ӳˮ����ˮ�ļ��鷽��������ˮ��Ӳ�ȵķ��������з������
��3������ˮ���������Ԫ�غ���Ԫ�ع��ɣ�������������ˮ�������
��� �⣺��1�������ǰѲ�����Һ��Ĺ�����Һ������һ�ַ�������ȥˮ�������Թ������ʵIJ����ǹ��ˣ�
��2��Ӳˮ����ˮ���������������ĸ�þ���ӵĶ��٣����÷���ˮ������Ӳˮ����ˮ���������ˮ����������ĭ�϶࣬������ˮ����������ĭ���٣�����Ӳˮ�������г��õĽ���ˮ��Ӳ�ȵķ�������У�
��3��ˮ���������Ԫ�غ���Ԫ�ع��ɣ�������������ˮ������ˮ����Ԫ�غ����ܵͣ������ṩ���������������ˮ������ı�����Դ��������ˮ��Դ��Ҫ��Լ��ˮ����Ҫ��ֹˮ����Ⱦ��
�ʴ�Ϊ����1�����ˣ���2������ˮ����У���3��B��
���� �����ѶȲ������չ��˵�ԭ����Ӳˮ����ˮ�ļ��鷽����ת����������ȷ�����Ĺؼ���

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ
8��ij������ܺ���H2��N2��CO��CH4�е�һ�ֻ����֣��������������������г��ȼ�պ�ȫ������������ͨ����������ˮ����ͭ������������Һ��������ˮ����ͭ����ɫ������������Һ���������ӣ��������ɷֲ������ǣ�������
| A�� | CO��CH4 | B�� | H2��N2 | C�� | CH4 | D�� | H2��CO |
12��ij��Ӧ����ʾ��ͼ���£�һ��С�����һ��Ԫ�ص�ԭ�ӣ�������˵����ȷ���ǣ�������
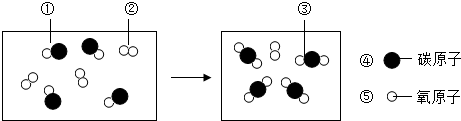

| A�� | �÷�Ӧ�����ֲ��� | B�� | �÷�Ӧ�����������غ㶨�� | ||
| C�� | �÷�Ӧ�еķ�Ӧ�ﶼ�ǻ����� | D�� | �÷�Ӧ���ڻ��Ϸ�Ӧ |
2�����������û���Ӧ���ǣ�������
| A�� | ��Ư��2Cl2+2Ca��OH��2=CaCl2+Ca��ClO��2+2H2O | |
| B�� | DZͧ����2Na2O2+2H2O=4NaOH+O2�� | |
| C�� | ��¯����3CO+Fe2O3$\frac{\underline{\;����\;}}{\;}$2Fe+3CO2 | |
| D�� | ��������2Al+Fe2O3$\frac{\underline{\;����\;}}{\;}$2Fe+Al203 |
9���ס������ֹ�����ܽ��������ͼ��ʾ������˵��������ǣ�������
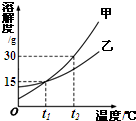

| A�� | ����t1��ʱ�����ܽ�ȴ����ҵ��ܽ�� | |
| B�� | t1��ʱ���ס��ұ�����Һ�����ʵ�����������Ϊ15% | |
| C�� | t2��ʱ�����ס��ҵı�����Һ������t1�棬�������Ĺ���һ�������� | |
| D�� | t2��ʱ����30 g�ס����зֱ����100 gˮ��������Һ�����ʵ�����������һ�������� |
6�����б仯�У������˻�ѧ�仯���ǣ�������
| A�� | ľ�������� | B�� | ���ڻ���ˮ | C�� | ����ȼ�� | D�� | ʪ�·����� |
7����ȷ�Ļ�ѧʵ�������ʵ������������ȫ�dz���Ҫ����ͼ�в�����ȷ�ģ�������
| A�� |  ��������ζ | B�� |  ȡ�ÿ�״���� | C�� | 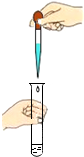 �μ�Һ�� �μ�Һ�� | D�� | 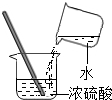 ϡ��Ũ���� |