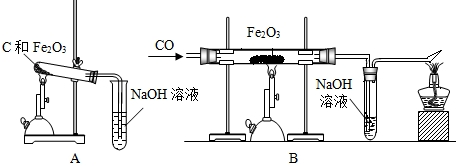
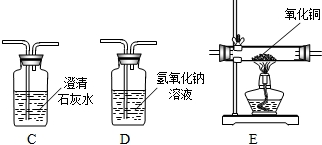
��Ŀ����
Ϊ�˲ⶨijʯ��ʯ��̼��Ƶ������������������ʲ��μӷ�Ӧ���Ҳ�����ˮ����ij��ȤС���������ʵ�鷽�����ٳ�ȡ10gʯ��ʯ��Ʒ���ڸ����������������ٸı䣬�۰�ʣ�������ڿ�������ȴ�����£��ܳƵ�ʣ����������Ϊ6.04g�����ڷ����в���������̼������Ϊ______g�������ʵ���Ϊ______mol��
�ڸ��ݷ�������ʯ��ʯ��̼��Ƶ��������������ݻ�ѧ����ʽ��ʽ���㣩��
��ָ����ʵ�鷽����һ�����ԵIJ��㲢˵��ԭ��______����
���𰸡��������ٸ��������غ㶨�ɿ�֪���������ļ������������ɶ�����̼���������ö�����̼����������������Է��������������ʵ������ڸ��ݶ�����̼�����������̼��Ƶ��������ٳ���ʯ��ʯ���������ɣ�����Ϊ�����ƾ�����ˮ�����Բ����ڿ�������ȴ��
����⣺���ڷ����в���������̼������Ϊ��10g-6.04g=3.96g�������ʵ���Ϊ��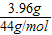 =0.09mol��
=0.09mol��
����Ҫ����3.96g ������̼��Ҫ̼�������ΪX��
CaCO3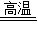 CaO+CO2��
CaO+CO2��
100 44
X 3.96g
���ݣ� ���X=9g��ʯ��ʯ��̼��Ƶ�����������
���X=9g��ʯ��ʯ��̼��Ƶ�����������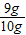 ×100%=90%��
×100%=90%��
����Ϊ�����ƾ�����ˮ�����Բ����ڿ�������ȴ��������������ˮ��Ӧ�����������ƣ������������ٵ����ˣ������
�ʴ�Ϊ����3.96g��0.09����90%�����ڿ�������ȴ�������ƻ�����ˮ�������������ƣ�
�����������Ĺؼ��ǹ��������ļ����������ɶ�����̼�����������ݶ�����̼�����̼��Ƶ�������Ҫ��������ƾ�����ˮ�ԣ�
����⣺���ڷ����в���������̼������Ϊ��10g-6.04g=3.96g�������ʵ���Ϊ��
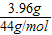 =0.09mol��
=0.09mol������Ҫ����3.96g ������̼��Ҫ̼�������ΪX��
CaCO3
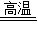 CaO+CO2��
CaO+CO2��100 44
X 3.96g
���ݣ�
 ���X=9g��ʯ��ʯ��̼��Ƶ�����������
���X=9g��ʯ��ʯ��̼��Ƶ�����������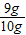 ×100%=90%��
×100%=90%������Ϊ�����ƾ�����ˮ�����Բ����ڿ�������ȴ��������������ˮ��Ӧ�����������ƣ������������ٵ����ˣ������
�ʴ�Ϊ����3.96g��0.09����90%�����ڿ�������ȴ�������ƻ�����ˮ�������������ƣ�
�����������Ĺؼ��ǹ��������ļ����������ɶ�����̼�����������ݶ�����̼�����̼��Ƶ�������Ҫ��������ƾ�����ˮ�ԣ�

��ϰ��ϵ�д�
�����Ŀ