ΧβΡΩΡΎ»ί
œ÷”– Β―ι “÷Τ»ΓΤχΧεΒΡ≤ΩΖ÷ΉΑ÷ΟΘ§«κΫαΚœΥυ―ßΜ·―ß÷Σ ΕΘ§ΜΊ¥π”–ΙΊΈ ΧβΘΚ
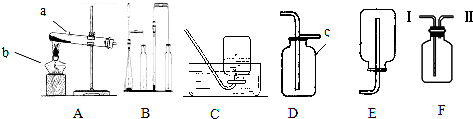
Θ®1Θ©«κ–¥≥ωΆΦ÷–±ξ”–Ή÷ΡΗΒΡ“«ΤςΟϊ≥ΤΘΚa_______ΘΜb_________ΘΜc__________ΓΘ
Θ®2Θ© Β―ι “”ΟΗΏΟΧΥαΦΊ÷Τ»Γ―θΤχΘ§ΉΑ÷ΟAΜΙ–ηΉωΒΡ“ΜΒψΗΡΕ· «____________Θ§«κ–¥≥ωΗΟΖ¥”ΠΒΡΜ·―ßΖΫ≥Χ ΫΘΚ___________________ΓΘ
Θ®3Θ© Β―ι “”ΟΙΐ―θΜ·«β»ή“Κ÷Τ»Γ―θΤχΘ§Ω…”ΟΒΡΖΔ…ζΉΑ÷Ο «_________Θ§«κ–¥≥ωΖ¥”ΠΒΡΜ·―ßΖΫ≥Χ ΫΘΚ_____________Θ§Τδ÷–Εΰ―θΜ·ΟΧΤπ_________Ής”ΟΓΘ»τ”ΟDΉΑ÷Ο ’Φ·―θΤχΘ§ ‘–¥≥ω―ι¬ζΒΡΖΫΖ®Θ®Αϋά®≤ΌΉςΓΔœ÷œσΓΔΫα¬έΘ©___________________________ΓΘ
Θ®4Θ© Β―ι “÷Τ»ΓΕΰ―θΜ·ΧΦΒΡΜ·―ßΖΫ≥Χ ΫΈΣΘΚ________________Θ§≥Θ”ΟΒΡΖΔ…ζΉΑ÷ΟΚΆ ’Φ·ΉΑ÷Ο «_______Θ§»τ“Σ ’Φ·ΒΫΗ…‘οΒΡΕΰ―θΜ·ΧΦΤχΧεΘ§”ΠΫΪΖΔ…ζΉΑ÷Ο”κFΉΑ÷ΟΒΡ________Θ®ΧνΓΑmΓ±ΜρΓΑnΓ±Θ©œύΝ§ΓΘ
Θ®5Θ©Ρή”ΟEΉΑ÷Ο ’Φ·ΒΡΤχΧε”–______________Θ®»ΈΨΌ“ΜάΐΘ©ΓΘ
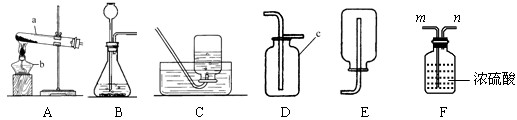
Θ®2Θ© Β―ι “”ΟΗΏΟΧΥαΦΊ÷Τ»Γ―θΤχΘ§ΉΑ÷ΟAΜΙ–ηΉωΒΡ“ΜΒψΗΡΕ· «____________Θ§«κ–¥≥ωΗΟΖ¥”ΠΒΡΜ·―ßΖΫ≥Χ ΫΘΚ___________________ΓΘ
Θ®3Θ© Β―ι “”ΟΙΐ―θΜ·«β»ή“Κ÷Τ»Γ―θΤχΘ§Ω…”ΟΒΡΖΔ…ζΉΑ÷Ο «_________Θ§«κ–¥≥ωΖ¥”ΠΒΡΜ·―ßΖΫ≥Χ ΫΘΚ_____________Θ§Τδ÷–Εΰ―θΜ·ΟΧΤπ_________Ής”ΟΓΘ»τ”ΟDΉΑ÷Ο ’Φ·―θΤχΘ§ ‘–¥≥ω―ι¬ζΒΡΖΫΖ®Θ®Αϋά®≤ΌΉςΓΔœ÷œσΓΔΫα¬έΘ©___________________________ΓΘ
Θ®4Θ© Β―ι “÷Τ»ΓΕΰ―θΜ·ΧΦΒΡΜ·―ßΖΫ≥Χ ΫΈΣΘΚ________________Θ§≥Θ”ΟΒΡΖΔ…ζΉΑ÷ΟΚΆ ’Φ·ΉΑ÷Ο «_______Θ§»τ“Σ ’Φ·ΒΫΗ…‘οΒΡΕΰ―θΜ·ΧΦΤχΧεΘ§”ΠΫΪΖΔ…ζΉΑ÷Ο”κFΉΑ÷ΟΒΡ________Θ®ΧνΓΑmΓ±ΜρΓΑnΓ±Θ©œύΝ§ΓΘ
Θ®5Θ©Ρή”ΟEΉΑ÷Ο ’Φ·ΒΡΤχΧε”–______________Θ®»ΈΨΌ“ΜάΐΘ©ΓΘ
(1) ‘ΙήΘΜΨΤΨΪΒΤΘΜΦ·ΤχΤΩ
(2)‘Ύ ‘ΙήΩΎ»ϊ“ΜΆ≈ΟόΜ®ΘΜ2KMnO4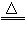 K2MnO4+MnO2+O2Γϋ
K2MnO4+MnO2+O2Γϋ
(3)BΘΜ2H2O2 2H2O+O2ΓϋΘΜ¥ΏΜ·ΘΜ”Ο¥χΜπ–«ΒΡΡΨΧθΖ≈‘ΎΦ·ΤχΤΩΩΎΘ§»γΙϊΡΨΧθΗ¥»ΦΘ§‘ρ÷ΛΟς―θΤχ“― ’Φ·¬ζΘ®Ζ¥÷°‘ρΟΜ”– ’Φ·¬ζΘ©
2H2O+O2ΓϋΘΜ¥ΏΜ·ΘΜ”Ο¥χΜπ–«ΒΡΡΨΧθΖ≈‘ΎΦ·ΤχΤΩΩΎΘ§»γΙϊΡΨΧθΗ¥»ΦΘ§‘ρ÷ΛΟς―θΤχ“― ’Φ·¬ζΘ®Ζ¥÷°‘ρΟΜ”– ’Φ·¬ζΘ©
(4)CaCO3+2HCl==CaCl2+H2O+CO2ΓϋΘΜBDΘΜm
(5)«βΤχΘ®ΜρH2Θ©ΓΔΘ®ΜρΦΉΆιΓΔΑ±ΤχΒ»ΟήΕ»±»Ω’Τχ–ΓΒΡΤχΧεΘ©
(2)‘Ύ ‘ΙήΩΎ»ϊ“ΜΆ≈ΟόΜ®ΘΜ2KMnO4
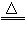 K2MnO4+MnO2+O2Γϋ
K2MnO4+MnO2+O2Γϋ(3)BΘΜ2H2O2
 2H2O+O2ΓϋΘΜ¥ΏΜ·ΘΜ”Ο¥χΜπ–«ΒΡΡΨΧθΖ≈‘ΎΦ·ΤχΤΩΩΎΘ§»γΙϊΡΨΧθΗ¥»ΦΘ§‘ρ÷ΛΟς―θΤχ“― ’Φ·¬ζΘ®Ζ¥÷°‘ρΟΜ”– ’Φ·¬ζΘ©
2H2O+O2ΓϋΘΜ¥ΏΜ·ΘΜ”Ο¥χΜπ–«ΒΡΡΨΧθΖ≈‘ΎΦ·ΤχΤΩΩΎΘ§»γΙϊΡΨΧθΗ¥»ΦΘ§‘ρ÷ΛΟς―θΤχ“― ’Φ·¬ζΘ®Ζ¥÷°‘ρΟΜ”– ’Φ·¬ζΘ©(4)CaCO3+2HCl==CaCl2+H2O+CO2ΓϋΘΜBDΘΜm
(5)«βΤχΘ®ΜρH2Θ©ΓΔΘ®ΜρΦΉΆιΓΔΑ±ΤχΒ»ΟήΕ»±»Ω’Τχ–ΓΒΡΤχΧεΘ©

ΝΖœΑ≤αœΒΝ–¥πΑΗ
 ‘ΡΕΝΩλ≥ΒœΒΝ–¥πΑΗ
‘ΡΕΝΩλ≥ΒœΒΝ–¥πΑΗ
œύΙΊΧβΡΩ