��Ŀ����
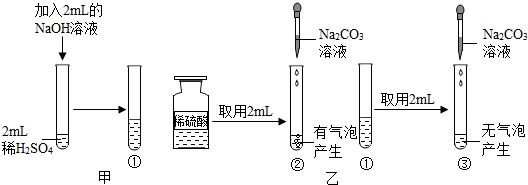
 ijͬѧ�ҵ���ˮ�ܵ������ˣ�����������һƿ���ܵ���ͨ����������˵����ͼ������ͨ����������������������Һ��Ӧʱ�ų��������ȣ��Լӿ�����������ë�����ٻ�������ã���Ӧ��ԭ��Ϊ��2Al+2NaOH+2H2O�T2NaAlO2+3H2������ش������й����⣺
ijͬѧ�ҵ���ˮ�ܵ������ˣ�����������һƿ���ܵ���ͨ����������˵����ͼ������ͨ����������������������Һ��Ӧʱ�ų��������ȣ��Լӿ�����������ë�����ٻ�������ã���Ӧ��ԭ��Ϊ��2Al+2NaOH+2H2O�T2NaAlO2+3H2������ش������й����⣺��1���ùܵ���ͨ����Ҫ�ܷⱣ���ԭ����
��2������ʹ��˵������ͬѧ�ԡ���ͨ�������˽�һ���о��������ձ��м���206gˮ���ټ��뱾Ʒ200g��������ȫ����Ӧ��������������1λС����
�������������������������=0.09g/L��
�ڷ�Ӧ����Һ���������Ƶ�����������
���㣺���ݻ�ѧ��Ӧ����ʽ�ļ���,�й��������������ļ���
ר�⣺�ۺϼ��㣨ͼ���͡������͡��龰�ͼ����⣩
��������1���ܵ���ͨ���е������ܺͿ����е�ˮ��Ӧ��Ϊ�˷�ֹʧЧ��Ӧ���ܷⱣ�棻
��2�����������������Լ����������������������ĵ��������Ƶ���������һ�����Լ�����������������ͷ�Ӧ����Һ���������Ƶ�����������
��2�����������������Լ����������������������ĵ��������Ƶ���������һ�����Լ�����������������ͷ�Ӧ����Һ���������Ƶ�����������
����⣺��1����Ϊ�������������ܺ�ˮ������ѧ��Ӧ���Ӷ����¹ܵ���ͨ��ʧЧ�����Ӧ���ܷⱣ�森
����׳��⣬��ˮ�����ʣ�
��2������������������Ϊx���μӷ�Ӧ���������Ƶ�����Ϊy��
2Al+2NaOH+2H2O�T2NaAlO2+3H2����
54 80 6
54g y x
=
=
��
x=6g��y=80g��
���������������
=66.7L��
��Ӧ��ʣ���������������Ϊ��200g-54g-80g=66g��
��Ӧ����Һ������Ϊ��206g+200g-6g=400g��
��Ӧ����Һ���������Ƶ���������Ϊ��
��100%=16.5%��
�����������������66.7L����Ӧ����Һ���������Ƶ�����������16.5%��
����׳��⣬��ˮ�����ʣ�
��2������������������Ϊx���μӷ�Ӧ���������Ƶ�����Ϊy��
2Al+2NaOH+2H2O�T2NaAlO2+3H2����
54 80 6
54g y x
| 54 |
| 54g |
| 80 |
| y |
| 6 |
| x |
x=6g��y=80g��
���������������
| 6g |
| 0.09g/L |
��Ӧ��ʣ���������������Ϊ��200g-54g-80g=66g��
��Ӧ����Һ������Ϊ��206g+200g-6g=400g��
��Ӧ����Һ���������Ƶ���������Ϊ��
| 66g |
| 400g |
�����������������66.7L����Ӧ����Һ���������Ƶ�����������16.5%��
������������Ҫ����ѧ�����ü��跨�ͻ�ѧ����ʽ���м�����ƶϵ�����������ʱҪע��淶�Ժ�ȷ�ԣ�

��ϰ��ϵ�д�
�����Ŀ
��������IJ�����������е�ˮ�����ص��ǣ�������
| A���ִ�ı��ɷ��ڿ����б��� |
| B�������δ�����IJ˽��˸е����ơ�ͷ�� |
| C������ӱ�����ȡ����ѩ��ð�������� |
| D��������糿�����������С������� |
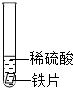
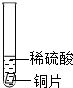
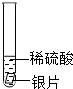
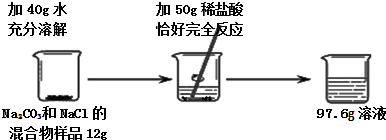
 С�����ֶ������̴���������ֽ��ʵ���У���Ӧ�ٶ��������������ϵ�֪����ԭ���ǹ����������������������С�Լ��������̵Ĵ��������ͣ�Ϊ��̽���������̴��������͵�Ӱ�����أ���������ʵ�飮
С�����ֶ������̴���������ֽ��ʵ���У���Ӧ�ٶ��������������ϵ�֪����ԭ���ǹ����������������������С�Լ��������̵Ĵ��������ͣ�Ϊ��̽���������̴��������͵�Ӱ�����أ���������ʵ�飮