��Ŀ����
���ſƼ��ķ�չ�����Ľ����������ѽ���Ѱ�����ռң�
��1����������������װ��Чת�����ɽ�����β�����ж�����ת��Ϊ�����壬��ͼ�Ǹû�ѧ��Ӧ���۹���ģ��ͼ����Ӧ�Ļ�ѧ����ʽΪ�� ��

��2�������Ҵ������ǽ��Ҵ������Ͱ�һ��������϶��ɵ�һ������ȼ�ϣ�
���Ҵ����� ��Դ������ţ���
A�������� B����������
�����ܱ������У��������Ҵ�����һ����������ȼ�գ��ֲ�÷�Ӧǰ������ʵ��������£�
����x��ֵΪ ��Wһ���� ��дһ�����ʵ����ƣ���
��3������վ�ر���������Ʒ֮һ��ɳ�ӣ���ɳ������ԭ���� ��
��4�������������ȹ�����ͨ����ʱ��������Ʒ��ֹЯ������ ������ţ���
A������ B��ƿװ���� C���鿯��־ D���ƾ���
��1����������������װ��Чת�����ɽ�����β�����ж�����ת��Ϊ�����壬��ͼ�Ǹû�ѧ��Ӧ���۹���ģ��ͼ����Ӧ�Ļ�ѧ����ʽΪ��

��2�������Ҵ������ǽ��Ҵ������Ͱ�һ��������϶��ɵ�һ������ȼ�ϣ�
���Ҵ�����
A�������� B����������
�����ܱ������У��������Ҵ�����һ����������ȼ�գ��ֲ�÷�Ӧǰ������ʵ��������£�
| ���� | �Ҵ� | ���� | ������̼ | ˮ���� | W |
| ��Ӧǰ����/g | 23 | 40 | 0 | 0 | 0 |
| ��Ӧ������/g | 0 | 0 | 22 | 27 | x |
��3������վ�ر���������Ʒ֮һ��ɳ�ӣ���ɳ������ԭ����
��4�������������ȹ�����ͨ����ʱ��������Ʒ��ֹЯ������
A������ B��ƿװ���� C���鿯��־ D���ƾ���
���㣺����ȼ�ϵ�ʹ������Ի�����Ӱ��,���۵㼰ģ��ͼ��Ӧ��,�����غ㶨�ɼ���Ӧ��,��д��ѧ����ʽ�����ֱ���ʽ�����뷽��ʽ,����ԭ���ͷ���,��ȼ����ױ��ﰲȫ֪ʶ,������Դ�����ࡢ��Դ�ķ���
ר�⣺��ѧ����Դ
��������1�����ݻ�ѧ��Ӧ���۹���ģ��ͼ��������Ӧ������P��Ӧ��������д����Ӧ�Ļ�ѧ����ʽ��
��2���ٸ����Ҵ����������ս��з�����
�ڸ��������غ㶨�ɽ��з�����
��3����������ԭ��������
��4��������ȼ�ױ���İ�ȫ֪ʶ�����жϣ�
��2���ٸ����Ҵ����������ս��з�����
�ڸ��������غ㶨�ɽ��з�����
��3����������ԭ��������
��4��������ȼ�ױ���İ�ȫ֪ʶ�����жϣ�
����⣺��1����ͼʾ�п��Կ���������������һ����̼�ڴ��������������ɶ�����̼�͵�������Ӧ�ķ���ʽ�ǣ�2NO2+4CO
4CO2+N2��
��2�����Ҵ��Ǿ���ֲ��ͺ����ɵģ����ڿ�������Դ
���������غ㶨�ɲμӷ�Ӧ�����������ܺ͵��������������֮�Ϳ�֪��x=23+40-22-27=14���ڻ�ѧ��Ӧ�У�ԭ�Ӹ�����ԭ������䣮���ԣ�Wһ����һ����̼��
��3����ɳ������ԭ���Ǹ���������
��4�����ڡ��ƾ�������ȼ�ױ���ڳ����������ȹ�����ͨ����ʱ��Ӧ��ֹЯ����
�ʴ�Ϊ����1��2NO2+4CO
4CO2+N2����2����A����14��һ����̼����3��������������4��AD��
| ||
��2�����Ҵ��Ǿ���ֲ��ͺ����ɵģ����ڿ�������Դ
���������غ㶨�ɲμӷ�Ӧ�����������ܺ͵��������������֮�Ϳ�֪��x=23+40-22-27=14���ڻ�ѧ��Ӧ�У�ԭ�Ӹ�����ԭ������䣮���ԣ�Wһ����һ����̼��
��3����ɳ������ԭ���Ǹ���������
��4�����ڡ��ƾ�������ȼ�ױ���ڳ����������ȹ�����ͨ����ʱ��Ӧ��ֹЯ����
�ʴ�Ϊ����1��2NO2+4CO
| ||
����������ͨ��������Ϊ����㣬�������뻯ѧ�йص�����֪ʶ�����ǿα��Ļ���֪ʶ��Ӧ��ǿ����Щ�����й�֪ʶ�Ĺ��ɺ�ѧϰ����߽�������������

��ϰ��ϵ�д�
�����Ŀ
����������������������̼��ƿ���壬��ѡ�õķ����ǣ�������
| A���������ǵ�ľ�����뵽����ƿ�� |
| B�����������ζ |
| C����ȼ�ŵ�ľ������ƿ�� |
| D���������ʯ��ˮ |
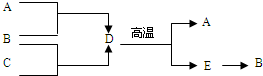 ABCDE�������ʾ�Ϊ���л�ѧ�еij������ʣ��������������ͼ��ʾ��ת����ϵ����֪A�ǹ�����õ�ԭ��֮һ��B�Ǹ��������������õļ��Իش��������⣺
ABCDE�������ʾ�Ϊ���л�ѧ�еij������ʣ��������������ͼ��ʾ��ת����ϵ����֪A�ǹ�����õ�ԭ��֮һ��B�Ǹ��������������õļ��Իش��������⣺ ijͬѧ��̽�����ۡ�˫�Ʒ��������ࡢ����塱��������۵���Ҫ�ɷ֣�
ijͬѧ��̽�����ۡ�˫�Ʒ��������ࡢ����塱��������۵���Ҫ�ɷ֣�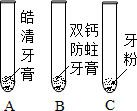
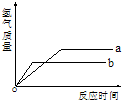 �����������ֽ���a��b�ֱ���������ϡ���ᷴӦ��������+2�۵Ļ�������������䷴Ӧ�����ͼ��ʾ��
�����������ֽ���a��b�ֱ���������ϡ���ᷴӦ��������+2�۵Ļ�������������䷴Ӧ�����ͼ��ʾ��