��Ŀ����
15�� ijƷ���������ı�ǩ��ͼ��
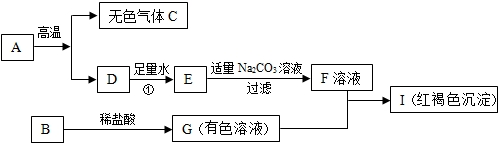
ijƷ���������ı�ǩ��ͼ����1�����������ƵĻ�ѧʽ��Na2PO3F������PԪ�صĻ��ϼ�Ϊ+5����FԪ�صĻ��ϼ�Ϊ-1����������Na��OԪ�ص�������Ϊ23��24��
��2������ˮ��Һ��ʹ��ɫ��̪��죬����ʼ��ԣ�����ԡ������ԡ����ԡ�����
��3������Ħ�����е�������CO32-���Ⱥ�����Ӧ�Ļ�ѧ����ʽΪCaCO3+2HCl=CaCl2+H2O+CO2�����÷�Ӧ�Ļ�������Ϊ���ֽⷴӦ��CO2+Ca��OH��2=CaCO3��+H2O��
��4��������������е��������Ƶ����������ﵽ0.76%��0.80%ʱ����ȣ�ݵ�Ч���Ϻã���ͨ�������жϸ�������У�����С������С����Ϻõķ�ȣ��Ч����
���� ��1�����ݻ��ϼ�ԭ���ɻ�ѧʽ���Ԫ�صĻ��ϼۣ����ݻ�ѧʽ���Ԫ�ص������ȣ�
��2������ָʾ���ı�ɫ������Һ������ԣ�
��3������̼������ӵļ��鷽������Ӧ���ص�����ش�
��4�����������е��������Ƶ��������������жϣ�
��� �⣺��1�����������ƵĻ�ѧʽ��Na2PO3F�������ƵĻ��ϼ�Ϊ+1�����Ļ��ϼ�Ϊ-2��PԪ�صĻ��ϼ�Ϊ+5����FԪ�صĻ��ϼ�Ϊx����+1����2+��+5��+��-2 ����3+x=0�����x=-1����������Na��OԪ�ص�������Ϊ����23��2������16��3��=23��24��
��2������ˮ��Һ��ʹ��ɫ��̪��죬����ʼ��ԣ�
��3������Ħ�����е�������CO32-�����ü���ϡ���ᣬ�ٽ����ɵ�����ͨ������ʯ��ˮ�������飬�Ⱥ�����Ӧ�Ļ�ѧ����ʽΪ CaCO3+2HCl=CaCl2+H2O+CO2�����÷�Ӧ�Ļ�������Ϊ���ֽⷴӦ��CO2+Ca��OH��2=CaCO3��+H2O��
��4��������������FԪ�ص���������Ϊ��$\frac{19}{23��2+31+16��3+19}$��100%=$\frac{19}{144}$��100%��13.19%����114mg=0.114g���ĵ��������Ƶ�����Ϊ0.114��13.19%��0.864g��
�����е��������Ƶ���������Ϊ$\frac{0.864g}{110g}$��100%��0.79%��
������Ϊ0.79%��0.76%��0.80%֮�䣬���нϺõķ�ȣ��Ч����
�ʴ�Ϊ����1��-1��23��24����2�����ԣ���3��CaCO3+2HCl=CaCl2+H2O+CO2�������ֽ⣬CO2+Ca��OH��2=CaCO3��+H2O����4�����У�
���� ������Ҫ�����˻�ѧ�������д���Լ����ݻ�ѧʽ�ļ���ȣ����ڿα��Ļ���֪ʶ��Ӧ��ǿѧϰ��

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�| A�� | ���� | B�� | ľ̿ | C�� | þ�� | D�� | ��� |
| �� �� | ѡ���Լ� | ��Ҫ���� | |
| A | CO2��CO�� | O2 | ��ȼ |
| B | MnO2��KCl�� | H2O | ���� |
| C | NaCl��Һ��CuCl2�� | AgNO3��Һ | �ᾧ |
| D | Fe��Cu�� | H2SO4 | ���� |
| A�� | A | B�� | B | C�� | C | D�� | D |
| A�� | �����к�������Ԫ���ǵ�Ԫ�� | |
| B�� | �ؿ��������������Ľ���Ԫ������Ԫ�� | |
| C�� | ���к���Ԫ�� | |
| D�� | ���к��⡢��Ԫ�� |
| A�� | ������͡����Է�������Ϊ407g | |
| B�� | ������͡��̼Ԫ������������� | |
| C�� | ������͡����43��ԭ�� | |
| D�� | ������͡�е�����Ԫ�ص�������Ϊ5�s1 |