��Ŀ����
ijУ��ѧ�о���ѧϰС��ͨ��ʵ���о���¯�����Ļ�ѧԭ�����������ͼ1ʵ��װ�ã�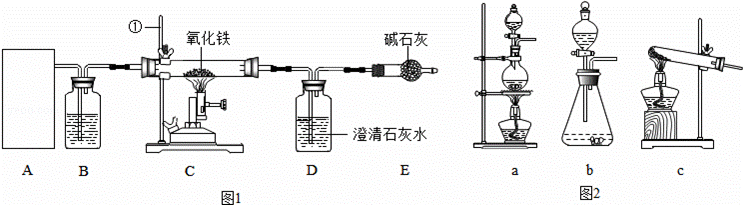
��1����֪HCOOH CO��+H2O�����ݸ÷�Ӧ��ȡCO��A��Ӧѡ�õ����巢��װ������������ͼ2������ĸ��ţ���
CO��+H2O�����ݸ÷�Ӧ��ȡCO��A��Ӧѡ�õ����巢��װ������������ͼ2������ĸ��ţ���
��2��װ���������ٵ�������������װ��B����ʢ�ŵ��Լ���������Eװ������Ϊ������
��3��ʵ�鿪ʼʱӦ�ȵ�ȼ�������A����C����������Դ����Ӧ��Cװ�õ�����������Dװ���з�����Ӧ�Ļ�ѧ����ʽΪ������Ӳ�ʴֲ�������������ȴ����ֹͣͨCO��Ŀ����������
��4����װ����һ�����Ե�ȱ�����������Ľ��ķ���Ϊ������
�⣺��1����Ӧ��ΪҺ�壬����Ϊ���ȣ���A��Ӧѡ�õ����巢��װ����a��
��2��װ���������ٵ�����������̨��װ��B����ʢ�ŵ��Լ�Ӧ���и������ã���Ũ���Eװ������Ϊ��ֹ�����еĶ�����̼����Dƿ��
��3��ʵ�鿪ʼʱӦ�ȵ�ȼ A������Դ������һ����̼��������ͨ��Cװ�ã���Dװ���ж�����̼�����ʯ��ˮ��Ӧ����̼��Ƴ�����ˮ����Ӧ�Ļ�ѧ����ʽΪ��CO2+Ca��OH��2�TCaCO3��+H2O����Ӳ�ʴֲ�������������ȴ����ֹͣͨCO��Ŀ����Ϊ�˷�ֹ����ʯ��ˮ�����벣������ʹ���ȵIJ�����ը�ѣ�
��4��ʵ������һ����һ����̼�ͷŵ��˿����У�����һ����̼�ж�������Ⱦ��������˱���Ҫ��ʵ�������β�����д���������������β�����е�ȼ���ռ��������ɣ�
�ʴ�Ϊ����1��a
��2������̨��Ũ�����ֹ�����еĶ�����̼����Dƿ��
��3��A����ɫ�����Ϊ��ɫ��CO2+Ca��OH��2�TCaCO3��+H2O��Ϊ�˷�ֹ����ʯ��ˮ�����벣������ʹ���ȵIJ�����ը�ѣ�
��4��ȱ��β������װ�ã���β�����е�ȼ

 ����ȫ���ִʾ��ƪ��ϵ�д�
����ȫ���ִʾ��ƪ��ϵ�д�ʵ���ҿ����ü�ʽ̼��ͭ����ѧʽ��Cu2��OH��2CO3���Ʊ�����ͭ��������̼�ۻ�ԭ����ͭ��ʵ�飮
���Ʊ�����ͭ��������װ����ͼ��ʾ��ʡ�Լг���������
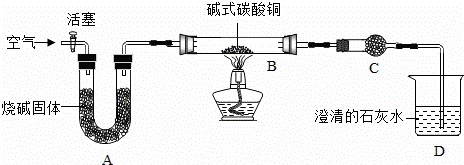
����һ������A��B��������ͨ�������
��������رջ���������C��D����ʼ���ȣ�
���������ڼ��ȹ����У���¼��B�й��������仯���±�����t3ʱֹͣ���ȡ�
| ����ʱ�䣨min�� | 0 | t1 | t2 | t3 |
| B���������g�� | 6.66 | 5.20 | 4.80 | 4.80 |
B�з�����Ӧ�Ļ�ѧ����ʽΪ����Cu2��OH��2CO3�T��CuO+��H2O+��CO2��
���ڡ���������ƽ���ϵ��������Ϊ �� �� ��
C���Լ��� ��ˮ����ͭ��A�����������տ����е� ˮ�����Ͷ�����̼��
��ֹ�Բ��������ɸ��ţ�ѡ��t3ʱֹͣ���ȵ�������
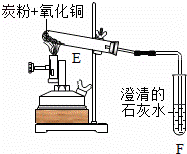 ��һ����̼�ۻ�ԭ�����Ƶõ�����ͭ��������װ����ͼ��ʾ��
��һ����̼�ۻ�ԭ�����Ƶõ�����ͭ��������װ����ͼ��ʾ��
ʵ������У��۲쵽F����Һ����ǣ�E�е�����
��
��Ӧ��ȫ���������߾ƾ����ֹͣ���ȣ����ܵ��µĺ��
��
�۷�����������ʡ�Բ��������Ԣ��з�Ӧ�����жϿ��ܲ�����
Ӱ�켰��ԭ��
���ݽ������˳��Fe��Cu��Ag�жϣ�����˵��������ǣ�������
| �� | A�� | ��ӦCu+FeSO4�TFe+CuSO4���Է��� |
| �� | B�� | ��ӦFe+2AgNO3�T2Ag+Fe��NO3��2���Է��� |
| �� | C�� | ͭƬ������������Һ�У���ɫ����ɫ�����ɫ |
| �� | D�� | ��˿��������ͭ��Һ�У���˿�����к�ɫ�������� |
��ֱ�ʢ�мס��ҡ������ֽ������Թ��еμ�ϡ���ᣬ���ס����ܲ����������ֱ�����X��Y�����ܴ�X��Һ���û����ף������ߵĽ������˳��Ϊ��������
| �� | A�� | �ף��ң��� | B�� | �ң��ף��� | C�� | �����ף��� | D�� | �ף������� |
 ������������ٶȣ�
������������ٶȣ�

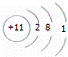 A��������ԭ�Ӻ�����11������
A��������ԭ�Ӻ�����11������