��Ŀ����
��ѧʵ���ǽ��п�ѧ̽������Ҫ��ʽ��
(1)����ʵ�������в������ڼ��ȵ���____������ĸ���ţ���
a���ձ� b����Ͳ c���Թ� d��������
(2)ij��ѧС��ѡ������װ�ú�ҩƷ����̽��ʵ�顣
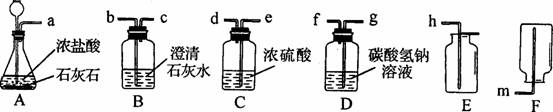 ��A�з�Ӧ�Ļ�ѧ����ʽΪ___________________________________________________��
��A�з�Ӧ�Ļ�ѧ����ʽΪ___________________________________________________��
������A��B���ӣ�A�в��������岢����ʹB�е���Һ����ǣ�����Ϊʲô��
����Ҫ��ȡһƿ��Ϊ�����������CO2����ѡ��װ�õĵ��ܽӿڴ����ҵ���ȷ����
˳��Ϊ��a��________��_________��________��________��________��
(3)Ϊ̽����ҵ��ˮ���ۺ����ã�ij��ѧС����ʵ���������������ʵ�顣
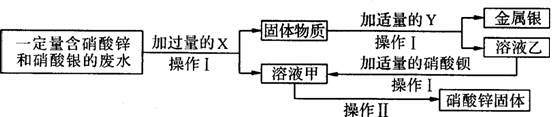 ��XΪ____���������ᱵ��Ӧ�Ļ�ѧ����ʽΪ_____________________________________��
��XΪ____���������ᱵ��Ӧ�Ļ�ѧ����ʽΪ_____________________________________��
�ڲ���I�Ͳ������ж�Ҫ�õ�һ�ֲ����������������ڲ������е�������ʲô��
(4)ij�������Ϊ����̽���÷����������ȡ�����Ʒ�����з�����4.9 t��H2SO4��
��������Ϊ20%��������������м��Ӧ����ȡ����������ͬʱ�����ɵ�ȫ������ͨ������
����ͭ�в����ȣ�H2+CuO == Cu +H2O�����������������������ͭ��������
(1) b ��1�֣�
(2)��CaCO3 +2HCl == CaCl2 + CO2�� + H2O (1��)
��Ũ�����ӷ���ʹCO2�л���HC1����������CaCO3������ ��1�֣�
|

 ��ĩ�����ϵ�д�
��ĩ�����ϵ�д�������ĸ A ~ H ��ʾ���л�ѧ���������ʣ��������⡢̼�������ȡ��ơ��ơ���
�е� 2��3 ��Ԫ����ɣ�
|
��2����ɫ��ĩ B ������ C ��Ӧ�õ���ɫ��ĩ����ѧ���� ʽΪ ��
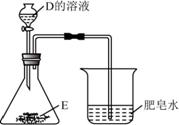
��3��D �� E ��������Ԫ�أ�����ͼ��ʾ���� D ����Һ��
����ƿ�У���Һ��ƣ������ݲ������ձ��оۼ��� �������ܱ���ȼ��E �к��е�Ԫ���� ��
��ƿ�з�����Ӧ�Ļ�ѧ����ʽΪ ��
��4��F��G��H �ֱ��� D ����Һ��Ӧ������ˮ���ɣ��� F ����Һ�� G ����Һ��ϣ����� ��ɫ���� H��F �� G ��Ӧ�Ļ�ѧ����ʽΪ ��
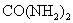 )��һ�ֵ��� B.���ʶ����ũ�����������Ҫ����
)��һ�ֵ��� B.���ʶ����ũ�����������Ҫ���� ��t1��ʱ��������Һ�����ʵ����������Ĵ�С��ϵ�� ��
��t1��ʱ��������Һ�����ʵ����������Ĵ�С��ϵ�� ��