��Ŀ����
12�����м��ּ�������������ˮ���ܼ����ƶ��ɵģ��书�ܼ���Ч�ɷ������ʾ���ݱ��ش��������⣺| �������� | ����� | ������Ư | Ư�� |
| ���� | ��Ч����۹�������ζ | Ưϴ���ʹɫ�ʸ����� | ����Ư���������� |
| ��Ч�ɷ� | HCl | H2O2 | NaClO |
��2��NaClO����Ԫ�صĻ��ϼ�Ϊ+1��
��3����ʢ��������������Ư��Һ����Թ��У��������������̣��۲쵽�������������ݲ�����
��4��������顱�롰Ư�������ܻ��ã�����������ײ����ж���������ͬʱ���Ȼ��ƺ�ˮ���ɣ��÷�Ӧ�Ļ�ѧ����ʽΪ2HCl+NaClO=NaCl+H2O+Cl2����
���� �������е����ʵ����ʽ��з�������������ʵ�pHС��7���ڻ������У���Ԫ���������ϼ۵Ĵ�����Ϊ�㣬�������ʼ䷴Ӧ�������Լ����ʼ䷴Ӧ��ѧ����ʽ����д���н�ɣ�
��� �⣺��1�����������ԣ�pHС��7�����С�ڣ�
��2����NaClO�У���Ԫ�صĻ��ϼ�Ϊ+1����Ԫ�صĻ��ϼ�Ϊ-2������Ԫ�صĻ��ϼ�Ϊx�����У���+1��+x+��-2��=0��x=+1�����+1��
��3����������Ư��Һ���к��й������⣬�ڶ������̵Ĵ��������������������ʻ�۲쵽�����ݲ�������������ݲ�����
��4����������������Ʒ�Ӧ�����Ȼ��ơ�ˮ�����������2HCl+NaClO=NaCl+H2O+Cl2����
���� ���⿼����dz�����������ʣ���ɴ��⣬�����������е�֪ʶ���У�

��ϰ��ϵ�д�
�����Ŀ
3��ijѧУ��չ�����¡���Һ����Եļ��顱��ʵ����
��1�������ƵĴ�컨֭��ͼ1��ʾ�ֱ�μӵ��Թ��У��۲쵽�����������ʾ��
��ͼ1��컨֭���ԣ�����ԡ������ԡ��������ָʾ����
�ھ��ϱ��Ʋ⣬��컨֭����ϡ������Һ��ʾ����ɫ�dz�ɫ��
��ȡ�����������������Թ��У������컨֭��������Һ��Ϊ��ɫ���ó����������Լ��ԣ�
��2����pH��ֽ���̼�����ϵ�pH���������������������=����7��
��3��ͼ2����λͬѧ�ֱ�ⶨ����ˮ����ԵIJ���ʾ��ͼ��������ȷ����C���A������B������C������ͬ�������ܵ��·���ˮpH��ֵ�����ı�Ĵ��������B��

��1�������ƵĴ�컨֭��ͼ1��ʾ�ֱ�μӵ��Թ��У��۲쵽�����������ʾ��
| �Լ� | �״� | ϡ���� | ʳ��ˮ | ����ˮ | ʯ��ˮ |
| ����컨֭�����ɫ | ��ɫ | ��ɫ | �ۺ�ɫ | ��ɫ | ��ɫ |
�ھ��ϱ��Ʋ⣬��컨֭����ϡ������Һ��ʾ����ɫ�dz�ɫ��
��ȡ�����������������Թ��У������컨֭��������Һ��Ϊ��ɫ���ó����������Լ��ԣ�
��2����pH��ֽ���̼�����ϵ�pH���������������������=����7��
��3��ͼ2����λͬѧ�ֱ�ⶨ����ˮ����ԵIJ���ʾ��ͼ��������ȷ����C���A������B������C������ͬ�������ܵ��·���ˮpH��ֵ�����ı�Ĵ��������B��
20���ҹ���ѧ����������Ϊ�����������أ�C15H22O5��--һ����������ű����ҩ������ȫ���ر��Ƿ�չ�й��ҵ��������˵������������2015��ŵ��������ѧ��ҽѧ�������й��������ص�˵��������ǣ�������
| A�� | �����ص���Է�������Ϊ282 | |
| B�� | �����غ�̼��ԼΪ63.8% | |
| C�� | ��������C��HԪ����������90��11 | |
| D�� | �����ط�����̼Ԫ�ء���Ԫ�ء���Ԫ����� |
17����һ��������AgNO3��Cu��NO3��2�Ļ����Һ�м���һ��������п�ۣ���ַ�Ӧ����ˣ���ҺΪ��ɫ����������ʵ����������жϣ�����˵���У���ȷ���ǣ�������
| A�� | ��Һ��һ��������AgNO3 | B�� | ������һ�����е���Cu | ||
| C�� | ������һ�����е���Ag | D�� | ��Һ��������Ŀ�������Cu2+ |
1��2015��8��12������������Σ�ջ�ѧƷ�Ѷⷢ�����֣���������ը������˾����Ա�������Ʋ���ʧ������Ա���ʱ���֣���ˮ������ѧƷ��ʯ��CaC2��ʱ�����˸����ҵı�ը��ԭ���Ƿ����˷�Ӧ��CaC2+2H2O�TCa��OH��2+X����X���������ȼ�ղ����ܱ�ը�������й�˵����ȷ���ǣ�������
| A�� | X�ǻ�ѧʽΪC2H2 | |
| B�� | ������Ӧ�����û���Ӧ | |
| C�� | ����������һ�������� | |
| D�� | CaC2�и�Ԫ�غ�̼Ԫ�ص�������Ϊ1��2 |
2�������С�ˮ���ֵ����ʲ�������Һ���ǣ�������
| A�� | ����ˮ | B�� | ��Ȫˮ | C�� | ʯ��ˮ | D�� | Ӳˮ |
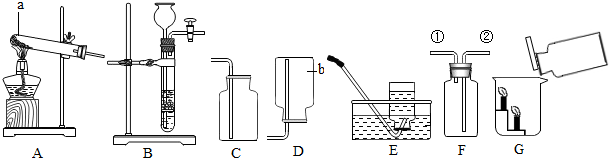
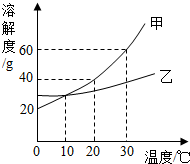 ��ͼ��ʾ�ļס������ֹ������ʵ��ܽ�����ߣ��ش�
��ͼ��ʾ�ļס������ֹ������ʵ��ܽ�����ߣ��ش�