��Ŀ����
1����ѧʵ�鳣�������Ե�ʵ�������������������ᷴӦ����������Ϊ��֤����������������ȷʵ�����˻�ѧ��Ӧ��ijͬѧ����������ʵ�飺��1��ȡ2mlNaOH��Һ���Թ��У�����1��2�η�̪�Լ�����Ȼ����μ���ϡ���ᣬͬʱ�������Թܣ��۲쵽����������Һ����ɫ��Ϊ��ɫ�����ɺ�ɫ��Ϊ��ɫ��
��2������ʵ������֤��������ϡ����ĵ��룬��Һ���Լ��������ǿ�������������������䡱�������������������������ᷢ�����кͷ�Ӧ���ɴ�֤��������ʵ�����ȷʵ�����˻�ѧ��Ӧ��
���� ��1����̪�Լ������죬�����Ժ�������Һ�г���ɫ������ϡ�����ϡ�����ܹ�������������Һ��Ӧ��ʹ��Һ�����ԣ����Ծݴ˽����⣻
��2��ϡ������������Ʒ�Ӧ�����������ƺ�ˮ��
��� �⣺��1������������Һ�ܹ�ʹ��̪��죬���Խ���̪��������������Һ�к��۲쵽��Һ��죬����̪�����Ժ�������Һ�г���ɫ�����Ե���μ��˷�̪������������Һ�еμ�ϡ����ᷢ����Һ�ĺ�ɫ���������ջ�����ɫ��
��2��ͨ���۲�ʵ����̿���֪��������ϡ����ĵ��룬��Һ������������������ϡ���������������Һ�������кͷ�Ӧ��
�ʴ�Ϊ����1����Һ����ɫ��Ϊ��ɫ�����ɺ�ɫ��Ϊ��ɫ��
��2��������
���� �����кͷ�Ӧ��ʵ��ʱ��������ͼ����Һһ������ɫ�����߷�Ӧʱ���������ԣ���������������һЩ�Լ��������ȣ������ָʾ����pH��ֽ���¶ȼƵȣ��������������жϷ�Ӧ�Ƿ���л�ǡ����ȫ��

��ϰ��ϵ�д�
 ��ҵ����ϵ�д�
��ҵ����ϵ�д� ͬ��ѧ��һ�ζ���ϵ�д�
ͬ��ѧ��һ�ζ���ϵ�д� �����ܾ�ϵ�д�
�����ܾ�ϵ�д� ���ƿ�����ϵ�д�
���ƿ�����ϵ�д�
�����Ŀ
11��C60��һ�����Ͳ��ϣ��㷺Ӧ���ڲ��Ͽ�ѧ��������ȷ��森C60���ڣ�������
| A�� | ����� | B�� | ���� | C�� | ������ | D�� | ������ |
12�����ݻ�ѧ����ʽCH4+2NO2�TX+CO2+2H2O�����ж�X�Ļ�ѧʽ�ǣ�������
| A�� | CO | B�� | O2 | C�� | N2 | D�� | H2 |
9����ͼ��A��B���ֹ������ʵ��ܽ����������˵���д�����ǣ�������


| A�� | t1��ʱA��B���ܽ����� | |
| B�� | t2��ʱ��A�ı�����Һ�����ʵ���������Ϊ$\frac{a}{100}$��100% | |
| C�� | A��B���ܽ�ȶ����¶����߶����� | |
| D�� | ��t2��ʱB�ı�����Һ���µ�t1��ʱ����Һ��ϡ�� |
6����Һ��������������������Ҫ�����壮
��1������ۡ�ʳ�Ρ����͡���4�����ʷֱ����ˮ�У����γ���Һ����ʳ�Σ�
��2��ijͬѧ��һ�������£���������Ϊ100g���ķ�ˮ�зֱ����һ������������أ�����������ܽ⣨���в��ܽ������أ������ȥ�����õ���Һ����ѧ����ʵ���м�¼���������ʾ
�����������ݷ�����ʵ��3��4����ʵ����ţ��õ�����Һ���ڱ�����Һ�����¶�������ص��ܽ����110g��
��3��Ϊ������50g��������Ϊ6%���Ȼ�����Һ����������²�����
�ټ��㣺����50g��������Ϊ6%���Ȼ�����Һ���Ȼ���3g��ˮ47g
�ڳ�������������ƽ���������Ȼ���ʱ������������ƽ��ָ��ƫ�����̣�ӦD��������ĸ��
A������ƽ����ĸ B����������������C�������������Ȼ��ƹ��� D�������Ȼ��ƹ���
����ȡ����50mL���10mL����20mL����50mL����
���ܽ⣺�����������ձ��У��ò��������裬ʹ�Ȼ����ܽ�
����Ҫʹ����Һ������������������һ����������������ȷ����C������ĸ��
A������10g�Ȼ��ƾ��� B������20g�Ȼ��ƾ���C��������25gˮ D��������50gˮ
��4����32.5gп��245gϡ����ǡ����ȫ��Ӧ�������ϡ�������������������д��������̣�
��1������ۡ�ʳ�Ρ����͡���4�����ʷֱ����ˮ�У����γ���Һ����ʳ�Σ�
��2��ijͬѧ��һ�������£���������Ϊ100g���ķ�ˮ�зֱ����һ������������أ�����������ܽ⣨���в��ܽ������أ������ȥ�����õ���Һ����ѧ����ʵ���м�¼���������ʾ
| ʵ����� | ʵ��1 | ʵ��2 | ʵ��3 | ʵ��4 |
| ˮ��������g�� | 100 | 100 | 100 | 100 |
| ��������ص�������g�� | 80 | 100 | 120 | 140 |
| ��Һ��������g�� | 180 | 200 | 210 | 210 |
��3��Ϊ������50g��������Ϊ6%���Ȼ�����Һ����������²�����
�ټ��㣺����50g��������Ϊ6%���Ȼ�����Һ���Ȼ���3g��ˮ47g
�ڳ�������������ƽ���������Ȼ���ʱ������������ƽ��ָ��ƫ�����̣�ӦD��������ĸ��
A������ƽ����ĸ B����������������C�������������Ȼ��ƹ��� D�������Ȼ��ƹ���
����ȡ����50mL���10mL����20mL����50mL����
���ܽ⣺�����������ձ��У��ò��������裬ʹ�Ȼ����ܽ�
����Ҫʹ����Һ������������������һ����������������ȷ����C������ĸ��
A������10g�Ȼ��ƾ��� B������20g�Ȼ��ƾ���C��������25gˮ D��������50gˮ
��4����32.5gп��245gϡ����ǡ����ȫ��Ӧ�������ϡ�������������������д��������̣�
13���������ʵ�ת�䣬������һ����Ӧʵ�ֵ��ǣ�������
| A�� | CuO����Cu��OH��2 | B�� | Ca��OH��2����NaOH | C�� | CO2����CaCO3 | D�� | MgCl2����Mg��NO3��2 |
10��Ϊ��ȥ���������е����ʣ�������Ϊ���ʣ���ѡ�õ��Լ��Ͳ�����������ȷ���ǣ�������
| ��� | �� �� | ѡ���Լ��������� | �������� |
| A | Cu��Fe�� | CuSO4��Һ | ����CuSO4��Һ����ַ�Ӧ����� |
| B | CO2��H2O�� | ������ | �������ͨ��ʢ�������Ƶ�U�� |
| C | NH3��H2O�� | Ũ���� | �������ͨ����Ũ�����ϴ��ƿ |
| D | NaOH��Һ[Ca��OH��2��Һ] | CO2���� | ͨ��CO2�����ַ�Ӧ����� |
| A�� | A | B�� | B | C�� | C | D�� | D |
11����ͼ��֪ʶ�Ǿ��꼶��ѧѧϰ����Ҫ֪ʶ����ش������й����⣺
��1������˵���в���ȷ����BD��������ĸ���ţ�
A����ͼ������Ԫ�ء�B�������κ�ˮ�ķ�Ӧһ�����кͷ�Ӧ
C��������ͼ��и�ʴ�� D����ͼ��������е��η������ֽⷴӦ
��2��Ϊ̽���ᡢ��Ļ�ѧ���ʣ�ijС��������ͼ��ʾʵ�飮
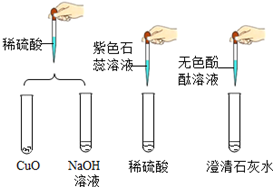
I��ʵ���ij�Թ���Ϊ��ɫ��Һ�������м���һ������ij��Һ��ɫ��ʧ��Ϊ��ɫ���÷�Ӧ�Ļ�ѧ����ʽΪ2HCl+Ca��OH��2�TCaCl2+2H2O��
II��ʵ��������õ�����ɫ��Һ����ɫ��Һ����ͬһ���ɾ����ձ��У��ɹ۲쵽����ɫ�������ɣ�������ԭ��2NaOH+CuSO4�TCu��OH��2��+Na2SO4
��3��ij���ڷ��õ�����������Һ�ѱ��ʣ��÷���ʽ��ʾ����ʵ�ԭ��2NaOH+CO2�TNa2CO3+H2O��
Ϊ֤����ƿ����������Һδ��ȫ���ʣ����������ʵ�飬�뽫������д������
��1������˵���в���ȷ����BD��������ĸ���ţ�
A����ͼ������Ԫ�ء�B�������κ�ˮ�ķ�Ӧһ�����кͷ�Ӧ
C��������ͼ��и�ʴ�� D����ͼ��������е��η������ֽⷴӦ
��2��Ϊ̽���ᡢ��Ļ�ѧ���ʣ�ijС��������ͼ��ʾʵ�飮

I��ʵ���ij�Թ���Ϊ��ɫ��Һ�������м���һ������ij��Һ��ɫ��ʧ��Ϊ��ɫ���÷�Ӧ�Ļ�ѧ����ʽΪ2HCl+Ca��OH��2�TCaCl2+2H2O��
II��ʵ��������õ�����ɫ��Һ����ɫ��Һ����ͬһ���ɾ����ձ��У��ɹ۲쵽����ɫ�������ɣ�������ԭ��2NaOH+CuSO4�TCu��OH��2��+Na2SO4
��3��ij���ڷ��õ�����������Һ�ѱ��ʣ��÷���ʽ��ʾ����ʵ�ԭ��2NaOH+CO2�TNa2CO3+H2O��
Ϊ֤����ƿ����������Һδ��ȫ���ʣ����������ʵ�飬�뽫������д������
| ʵ����� | ���� | ���� |
| ȡ��������Ʒ������������ CaCl2��Һ����ַ�Ӧ����ˣ� | �а�ɫ�������� | �йط�Ӧ�Ļ�ѧ����ʽ�� CaCl2+Na2CO3�TCaCO3��+2NaCl |
| ����Һ�У����뼸�� ��ɫ��̪��Һ | ��Һ��� | ��Ʒδ��ȫ���ʣ��Ժ����������ƣ� |