��Ŀ����
3����������Һ�õ�������ˮ���������÷���Ϊ0.9%���Ȼ�����Һ��ijУ��ȤС��ͬѧΪ�˼���ijһ������ˮ��Ʒ�Ƿ���ϱ�������������ʵ�飺��1����ȷ��ȡ�����������ˮ130ml�������ձ��У�130ml������ˮ�����ɽ�����130�ˣ���
��2�����ձ��м�����������������Һ���ٵμ�ϡ���
��3����������Һ���ˣ�Ȼ����ϴ�ӡ���ɣ���ȷ�������õ�����2.87�ˣ�
��������ʵ��ش��������⣺
��1�����˲�����ʹ�õIJ����������ձ�����������©����
��2��ͨ�����㣬�жϴ�������ˮ�Ƿ���ϱ�����Ҫ��д��������̣���
���� �Ȼ��ƺ���������Ӧ�����Ȼ��������������ƣ����ݷ�Ӧ�Ļ�ѧ����ʽ�����ṩ�����ݿ��Խ�����ط���ļ��㣮
��� �⣺��1�����˲�����ʹ�õIJ����������ձ�����������©����
���©����
��2�����Ȼ�������Ϊx��
NaCl+AgNO3�TAgCl��+NaNO3��
58.5 143.5
x 2.87g
$\frac{58.5}{x}$=$\frac{143.5}{2.87g}$��
x=1.17g��
�Ȼ�����������Ϊ��$\frac{1.17g}{130g}$��100%=0.9%��
ͨ�����㣬��������ˮ���ϱ���
�𣺸�������ˮ���ϱ���
���� ������Ҫ����ѧ�����ü��跨�ͻ�ѧ����ʽ���м�����ƶϵ�����������ʱҪע��淶�Ժ�ȷ�ԣ�

��ϰ��ϵ�д�
�����Ŀ
13���������ʣ���Fe2O3��BaCl2��Na2CO3���ܷ�����Ӧ���ǣ�������
| A�� | ˮ | B�� | ����ʯ��ˮ | C�� | ϡ���� | D�� | ��������Һ |
14��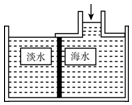 ��ˮ�����ɲ���Ĥ���뼼��������ͼ��ʾ���Ե���Ĥ�Ҳ�ĺ�ˮ��ѹ��ˮ���ӿ���������Ĥ���뵭ˮ�أ�����ˮ�еĸ������ʲ���ͨ������Ĥ���Ӷ��õ���ˮ���Լ�ѹ���Ҳຣˮ�ɷֽ��з�����������ǣ�������
��ˮ�����ɲ���Ĥ���뼼��������ͼ��ʾ���Ե���Ĥ�Ҳ�ĺ�ˮ��ѹ��ˮ���ӿ���������Ĥ���뵭ˮ�أ�����ˮ�еĸ������ʲ���ͨ������Ĥ���Ӷ��õ���ˮ���Լ�ѹ���Ҳຣˮ�ɷֽ��з�����������ǣ�������
 ��ˮ�����ɲ���Ĥ���뼼��������ͼ��ʾ���Ե���Ĥ�Ҳ�ĺ�ˮ��ѹ��ˮ���ӿ���������Ĥ���뵭ˮ�أ�����ˮ�еĸ������ʲ���ͨ������Ĥ���Ӷ��õ���ˮ���Լ�ѹ���Ҳຣˮ�ɷֽ��з�����������ǣ�������
��ˮ�����ɲ���Ĥ���뼼��������ͼ��ʾ���Ե���Ĥ�Ҳ�ĺ�ˮ��ѹ��ˮ���ӿ���������Ĥ���뵭ˮ�أ�����ˮ�еĸ������ʲ���ͨ������Ĥ���Ӷ��õ���ˮ���Լ�ѹ���Ҳຣˮ�ɷֽ��з�����������ǣ�������| A�� | ��ˮ������������������ | B�� | �ܼ��������� | ||
| C�� | ��ˮ�������� | D�� | ������������ |
11�����й�����Һ���ܽ�ȵ�˵���У�������ǣ�������
| A�� | ��������Һת��Ϊ������Һ����Һ�����ʵ������������ܱ��ֲ��� | |
| B�� | �¶Ƚ���ʱ��ԭ������Һ���ܲ��������� | |
| C�� | �¶�һ��ʱ������ص��ܽ�ȼ�����صı�����Һ�����ʵ�������������һ��ֵ | |
| D�� | ij��Һ�����������Һ�����ʵ���������һ����С |
18�������ǻ�ѧѧϰ�г��õ�˼ά����������������ȷ���ǣ�������
| A�� | ����Һ�ܸ�ijЩ�ǽ��������ﷴӦ�����Լ���Һ������CO���� | |
| B�� | ��������һ��Ԫ����ɵ����ʣ�����һ��Ԫ����ɵ�����һ���ǵ��� | |
| C�� | ��ѧ�仯�з���������ı䣬���Է���������ı�ı仯һ���ǻ�ѧ�仯 | |
| D�� | ����Һ��ʹ��ɫʯ����Һ��죬������ʹ��ɫʯ����Һ����һ��������Һ |
8�����и������ʣ����������Լ�����һһ����������ǣ�������
| A�� | Ba��NO3��2��NaOH��H2SO4��KCl | B�� | AgNO3��KCl��HNO3��MgCl2 | ||
| C�� | Ba��NO3��2��NaCl��CuSO4��NaOH | D�� | FeCl3��KOH��NaCl��NaNO3 |
12���ܶ�Ϊ0.91g/cm3�İ�ˮ����������Ϊ25%���ð�ˮ�õ������ˮϡ�ͺ�������Һ������������������
| A�� | ����12.5% | B�� | ����12.5% | C�� | С��12.5% | D�� | ��ȷ�� |
13��ijЩ�����⻯����ˮ��Ӧ�����ɼ���������� CaH2+2H2O�TCa��OH��2+2H2����NaH �� CaH2�Ļ�ѧ�������ƣ��� NaH ����������ϡ�����У����ɵ�������Ϊ��������
| A�� | NaOH �� H2 | B�� | NaOH �� NaCl | C�� | NaOH��H2�� NaCl | D�� | NaCl �� H2 |