��Ŀ����
���������;�㷺��ҽҩ�Ͽ�������������ɱ������ʵ����Ҳ����ʹ�ø��������ȡ�������밴Ҫ������������⣺
(1)������صĻ�ѧʽ_______��
(2)�����������Ԫ�صĻ��ϼ�___________��
(3)��������������ӵķ���______��
(4)��������������ӵķ���____________��
KMnO4+7K+MnO4- �������� (1)������صĻ�ѧʽKMnO4 (2)��������м�Ԫ�صĻ��ϼ���+1�ۣ���Ԫ�صĻ��ϼ�Ϊ-2�ۣ�����Ԫ�صĻ��ϼ�Ϊx���ݻ��ϼ۵Ĵ�����Ϊ0�����������Ԫ�صĻ��ϼ���+7�ۣ� (3)����������ɼ����Ӻ���������ӹ��ɣ��ʸ�������������ӵķ�����K+�� (4)����������ɼ����Ӻ���������ӹ��ɣ��ʸ�������������ӵķ���MnO4...(1)ij�о���ѧϰС��Ϊ��֤��������ԭ����ͭ����ʵ�飬����������ϲ�������������
�ʿ�����
I.������һ����ɫ���д̼�����ζ�����壬�ܶȱȿ���С����������ˮ����Һ�ʼ��ԡ�
II.������ԭ����ͭ�Ļ�ѧ��Ӧԭ��2NH3+3CuO 3Cu+N2+3H2O
3Cu+N2+3H2O
ʵ������
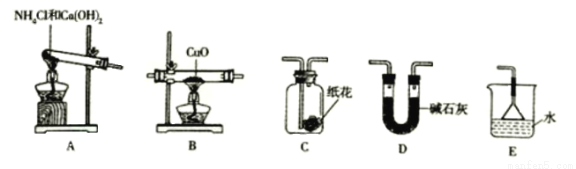
��ش�
��Ϊ��ɡ�������ԭ����ͭ����ʵ��Ŀ�ģ�װ��A��B��D��E����ȷ����˳��________ (�����)��
��װ��A�з�����Ӧ�Ļ�ѧ����ʽ_____________��
��װ��B�۲쵽������________��װ��E������_____________��
��װ��A����������ͨ��װ��C�У�ʯ����Һ���ݵ�ֽ�����ɫ��ԭ����________��
(2)С��ͬѧ�����Ȼ������������������Լ����������ơ�����þ���Ȼ�þ��ƿδ����ǩ����ɫ��Һ�м��������þ��Һ����Ʋ���������ʵ�顣���㲹��ʵ�����ݣ�
ʵ�鲽�� | ʵ������ | ʵ����� |
1.ȡ��֧�־��Թܣ��ֱ���������ɫ��Һ��Ȼ��μ� _________�����۲����� | ______________ | ______________ |
2. ______________ | ______________ | ______________ |




