��Ŀ����

̽��������ʵ���ҵķ�ˮ����ͭ���Ʊ������������塣
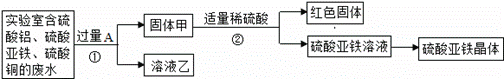
��1������ٵĻ�ѧ����ʽ�� ��
��2��������к��е������� ����ɫ������ ����Һ���е������� ��
̽���������������壨FeSO4•xH2O���Ļ�ѧʽ��
���������ϡ�
��1����ˮ����ͭ��ĩ��ˮ������ɫ������ͭ���塣
��2�����������������ʧȥ�ᾧˮ�����¼����ֽ�����������������̬�ǽ��������
��3���������������������Ե������ױ������������ӡ�
������ʵ�顿
����ȤС��ͬѧ��ȡag �����������壨FeSO4•xH2O����Ʒ��ͼ1װ�ø��¼���
�����������壨FeSO4•xH2O����Ʒ��ͼ1װ�ø��¼��� ��ʹ����ȫ�ֽ⣬�������ò�����з�����������SDTQ600�ȷ����Ƕ��������������ȷֽ���������ݣ����Ƴ�ͼ2��ʾ��ϵͼ��
��ʹ����ȫ�ֽ⣬�������ò�����з�����������SDTQ600�ȷ����Ƕ��������������ȷֽ���������ݣ����Ƴ�ͼ2��ʾ��ϵͼ��
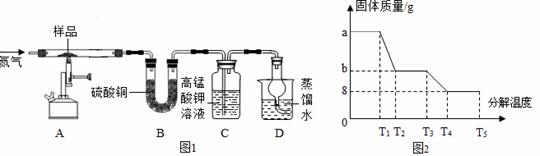
�����ݷ�����
��1��ͼ1װ��B����ˮ����ͭ��ĩ��������������12.6g��˵���������� ��
��2��ʵ����Ҫ����ͨ�뵪������������xֵ�ᣨ�ƫ��ƫС�����䣩 ��
��3��T4��ʱ�������������徭��ȫ�ֽ��װ��A�й���ʺ�ɫ����������ϡ���ᣬ����ȫ���ܽ⣬�õ���ɫ��Һ����װ��A�к�ɫ�����ǣ��ѧʽ�����ƣ� ��
��4�������FeSO4•xH2O�е�x����д��������̣�
���������ۡ�������������Ƭ�����³������ڰ�װҩ���⣬�����ܾ��е������� ��
��10�֣�������������ÿ��1�֣�
̽����1��Fe+CuSO4==FeSO4+Cu (2) Fe��Cu Cu Al2(SO4)3�� FeSO4
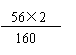 ̽����1��ˮ ��2��ƫС ��3��Fe2O3
̽����1��ˮ ��2��ƫС ��3��Fe2O3
��4����Ԫ�ص�����==8g�� =5.6g
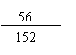
 5.6g��15.2g���һ������1�֣�
5.6g��15.2g���һ������1�֣�
��������������==5.6g�� =15.2g
FeSO4��XH2O
152 18X
15.2g 12.6g
X=7��1�֣�
���������ۡ�������������ֹҩƷ���ʣ��������ɣ�

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д���֪ ÿ�����ӵ�����ԼΪÿ�����ӣ������ӣ�������1/1836�������ڿ�ѧ�ϼ���ԭ�ӵ�����ʱֻ�������Ӻ����ӵ������������������Բ�
ÿ�����ӵ�����ԼΪÿ�����ӣ������ӣ�������1/1836�������ڿ�ѧ�ϼ���ԭ�ӵ�����ʱֻ�������Ӻ����ӵ������������������Բ� �ƣ��±��Dz���ԭ�ӵĹ���ָ����
�ƣ��±��Dz���ԭ�ӵĹ���ָ����
| ԭ������ | ������ | ������ | ��������� | ���ԭ������ |
| �� | 1 | 0 | 1 | 1 |
| ̼ | 6 | 6 | 6 | 12 |
| �� | 8 | 8 | 8 | 16 |
| �� | 11 | 12 | 11 | 23 |
| �� | 26 | 30 | 26 | 56 |
| �� | 92 | 146 | 92 | 238 |
ͨ���˱������ܽ������ԭ�������������ڵ��������������ܽ����
��1��_______________________________________________________________��
��2��_______________________________________________________________��
��3��___________________________________ ____________________________��
____________________________��
��4��_______________________________________________________________��
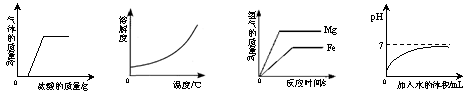
�����ĸ�ͼ��ӳ�˶�Ӧʵ�������������ı仯�����д������
|
| |||
| A���ڲ��ֱ��ʵ�NaOH��Һ�еμ�ϡ���� | B��O2���ܽ�����¶�Ӱ��ı仯���� | C����������Mg�ۺ�Fe��������ͬŨ�ȵ�ϡ���ᷴӦ | D��������Һϡ������pH�ı仯���� |
�±��г������ֽ���Ԫ�ص�ԭ�ӽṹ�����ʵ����ʵȾ��������
| Ԫ�� ���� | Ԫ�� ���� | ԭ�ӽṹʾ��ͼ | �ܶ� /g��cm��3 | �۵� /�� | �е� /�� | ����ˮ ��Ӧ |
| � | Li |
| 0.534 | 180.5 | 1 347 | ���� |
| �� | Na |
| 0.97 | 97.81 | 882.9 | ���� |
| �� | K |
| 0.86 | 63.65 | 774 | ���� �� ��ը |
| � | Rb |
| 1.532 | 38.39 | 688 | ���� �� ��ը |
�����������Ϸ������㷢������Щ���ɣ�
(1)________________________________________________________________________��
(2)________________________________________________________________________��
(3)________________________________________________________________________��
(4)�ϱ����й����У���һ�ֹ����Ǿ����������ɵģ��ù����ǣ�
________________________________________________________________________��
 ������ը
������ը