��Ŀ����
(8��)��ͼ��ʵ������������ȡ���ռ������װ��ͼ����ش��������⣺
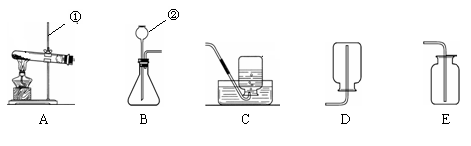
��1��д����ͼ�д�������������ƣ���__________����____________��
��2���ø��������ȡ����������װ��ѡ��_____������ĸ��ţ���ͬ������Ӧ�Ļ�ѧ����ʽΪ_______________________________________________��
��3��ʵ������ȡ������̼��Ӧѡ�õĹ���ҩƷ��_______���ռ�װ��ѡ��______����д���ó���ʯ��ˮ���������̼�Ļ�ѧ����ʽ____________________��
��4����ͼ��ijͬѧ�����������ϵ�һƿ���г�������ɫ�������壬����Ϊ�����������________��дһ�ּ��ɣ���

�� ����̨ ����©��
�� A 2KMnO4��K2MnO4 + MnO2 + O2��
�� ����ʯ����ʯ��ʯ�� E Ca(OH)2+CO2=CaCO3��+H2O
�� CO2 ����O2��
����:��

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ





