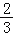��Ŀ����
ij��ѧ�ڶ�����С��������ѧ����֪ʶ�������ú�FeSO4��CuSO4�ķ�ˮ(��������������)�����л�������������壬����������ͼ��ʾ��
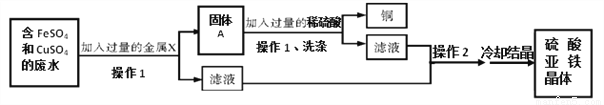
(1)���������У����������β���1������1��_____________(���������)��
(2)����X ��______(�ѧ����)��������ˮ������Ӧ�Ļ�ѧ����ʽΪ��____________������A�к���__________(�ѧ����)��д������A��ϡ���ᷴӦ�Ļ�ѧ����ʽΪ��____________________��
(3)ͨ����ȴ�ᾧ���õ�������������ǰ��Ҫ���в���2�����ʲ���2��________��
���� Fe Fe + CuSO4=Cu + FeSO4 Cu��Fe Fe + H2SO4 =FeSO4 + H2�� ����(��������Ũ��) �����������⿼���˽������˳����Ӧ�á� (1)����1�ǽ�������Һ����룬���Բ���1�ǹ��ˣ� ��2��Ϊ��ȥ��Һ�е�Cu2+���������ͭǰ��Ľ��������û���Ҫ�õ���Ϊ����FeSO4����Һ��ֻ�����Һ�м������������������ͭ�����û���Ӧ������...
��ϰ��ϵ�д�
�����Ŀ