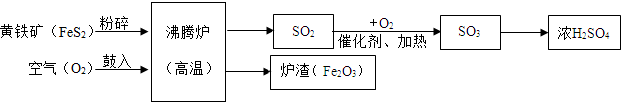
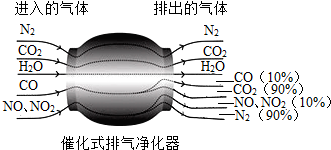
��Ŀ����
2����������ͼ���е���Ϣ��ѧϰ��ѧ��һ����Ҫ��������1�������KNO3��NaCl�ڲ�ͬ�¶��µ��ܽ�ȣ���λ��g/100gˮ��
| �¶ȣ��棩 | 0 | 10 | 20 | 30 | 40 | 50 | 60 | 70 | 80 | 90 | 100 |
| KNO3 | 13.3 | 20.9 | 32 | 45.8 | 64 | 85.5 | 110 | 138 | 169 | 202 | 246 |
| NaCl | 35.7 | 35.8 | 36 | 36.3 | 36.6 | 37 | 37.3 | 37.8 | 38.4 | 39 | 39.8 |
��.30��ʱ��KNO3��Һ�����Ũ�ȣ���������������Ϊ$\frac{45.8g}{45.8g+100g}$��ֻ�м���ʽ������Ҫ����������
��KNO3 �к������Ȼ���ʱ���ᴿ����ص�һ�㷽���ǽ��½ᾧ��
�������ݱ������ݿ�֪��KNO3��NaCl��ijһ�¶�ʱ������ͬ���ܽ��X����X��ȡֵ��Χ��36��36.3��
��2����ͼ�Ƕ�20��һ�������ļ���Һ���к��������ᾧ��ʵ����̣���ش�
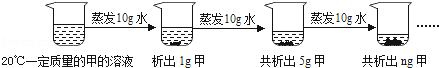
������ǰԭ��Һ�Dz����ͣ�����͡������͡�����Һ��n����ֵ��9��
����������ʵ�飬����Ϊ���ܽ�����¶ȱ仯��������C������ĸ����
A�����¶����߶����� B�����¶����߶���С C�����жϣ�
���� ��1����1�������ܽ�ȵĺ��塢������Һʱ�������������������ص��ܽ�����¶ȵ����߶�����KNO3��NaCl��20�桫30��ʱ������ͬ���ܽ�Ƚ��н��
��2������ݱ�����Һ�Ķ�����жϷ������з������������е����ݽ��з�����
�������������������ͨ�������ܼ��õ��Ľ��з�����
��� �⣺�⣺��1������ͼ����֪��20��ʱ��NaCl���ܽ����36g/100gˮ�����36��
��.30��ʱ��������Һʱ���������������30��ʱ������ص��ܽ��Ϊ45.8g��KNO3��Һ�����Ũ�ȣ���������������Ϊ$\frac{45.8g}{45.8g+100g}$��
������ص��ܽ�����¶ȵ����߶��������Դ�KNO3��Һ�еõ�KNO3��һ�㷽���ǽ��½ᾧ������ȴ�ᾧ��ȴ�ȱ�����Һ����
�������ݱ������ݿ�֪��KNO3��NaCl��20�桫30��ʱ������ͬ���ܽ�ȣ�����KNO3��NaCl��ijһ�¶�ʱ������ͬ���ܽ��X����X��ȡֵ��Χ��36��36.3��
��2��������ǰ���ձ��ײ�û�й��壬��һ������10gˮ������1g���壬�ڶ�������10gˮ������5g���壬ʵ�ʵڶ�����������4g���壬��������ǰԭ��Һ�Dz�������Һ������������10gˮ��4g���壬���Թ�����5g+4g=9g��
����������������ͨ�������ܼ��õ��ģ������жϳ��¶ȶ��ܽ�ȵ�Ӱ�죬��ѡ��C��
�ʴ�Ϊ����1����36����$\frac{45.8g}{45.8g+100g}$�����½ᾧ����36��36.3��
��2�������ͣ�9����C��
���� �����ؼ���Ҫ֪���ܽ�ȵĶ��壬�ܽ�����ߵ����壬֪��������Һ�������������ļ��㷽����

 ���¿쳵����������ϵ�д�
���¿쳵����������ϵ�д�| A�� |  ������Ϻ�� | B�� |  ��ׯ��� | C�� |  �㱱������ | D�� |  С����� |
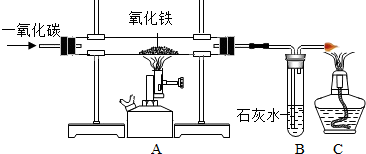
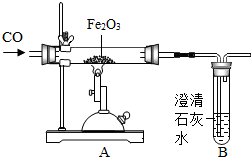 ��ͼΪ��һ����̼����������Ӧ����ʵ��װ�ã��÷�Ӧ����CO�Ļ�ԭ�ԣ���Fe2O3�е�����ԭ������
��ͼΪ��һ����̼����������Ӧ����ʵ��װ�ã��÷�Ӧ����CO�Ļ�ԭ�ԣ���Fe2O3�е�����ԭ������