��Ŀ����
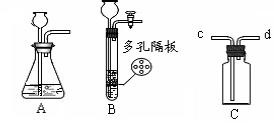
��13�֣�����ͼʾʵ��װ�ã��ش��������⡣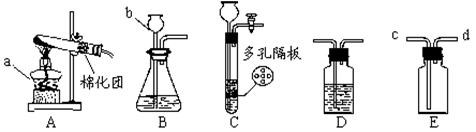
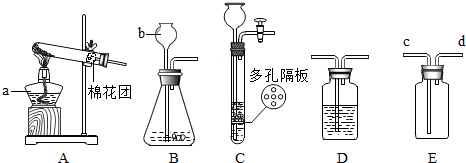
��1��ͼ��a��b���������ƣ�a ��b ��
��2���ø�����ع�����������ѡ�õķ���װ���� ���A������B����C������װ��ҩƷǰӦ�� ����Ӧ�Ļ�ѧ����ʽ�� ���÷�Ӧ�Ļ�������Ϊ ��Ӧ ��
��3��ijͬѧ��ʵ������Cװ�ã�����������ſ�״���壩��ȡ��������Ӧ�Ļ�ѧ����ʽΪ ���÷�Ӧ�Ļ�������Ϊ ��Ӧ��Cװ����Bװ����Ƚϣ��ŵ�Ϊ �����һ�㼴����
��4��ʵ������Bװ����ȡ������̼ʱ��b��Ӧ���� ����Ӧ�Ļ�ѧ����ʽΪ ����Ҫ��ø�������壬������װ���⣬��Ӧѡ��ʢ�� ����д�Լ����ƣ���Dװ�á�����Eװ���ռ�CO2��������Ӧ�� ��ͨ�루�c����d������
��1�� a. �ƾ��� b. ����©��
��2��A�� ���װ�������ԣ� 2KMnO4 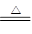 K2MnO4+ MnO2+O2�� �ֽⷴӦ
K2MnO4+ MnO2+O2�� �ֽⷴӦ
��3��Zn+H2SO4=ZnSO4+H2�� �û���Ӧ
������ʱ���Ʒ�Ӧ�ķ�����ֹͣ�����濪���á������ͣ��������������������˼��������֣�
��4��ϡ���ᣨ��ϡHCl�� CaCO3��2HCl=CaCl2��CO2����H2O
Ũ���ᣨ��ŨH2SO4�� c
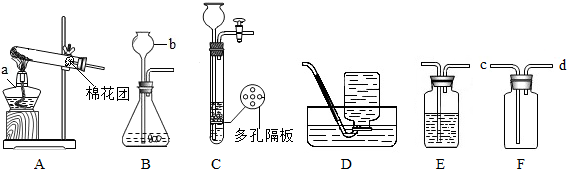
���������������1��a����Դ�������оƾ��ƣ�b�Ǽ���Һ��ҩƷ��������û�иǺͻ������г���©����
��2���ø�����ع����������ķ�Ӧԭ���Ǹ���������ȷֽ���������ء��������̡���������Ӧ����ʽΪ2KMnO4 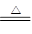 K2MnO4+ MnO2+O2�����ǷֽⷴӦ������ҩƷΪ���壬�����Ǽ��ȣ�����ҩƷ״̬�ͷ�Ӧ����������װ��ӦѡA����ȡ������ռ������ʵ�飬ʵ���ڼ���ҩƷǰҪ�ȼ��װ�õ������ԡ�
K2MnO4+ MnO2+O2�����ǷֽⷴӦ������ҩƷΪ���壬�����Ǽ��ȣ�����ҩƷ״̬�ͷ�Ӧ����������װ��ӦѡA����ȡ������ռ������ʵ�飬ʵ���ڼ���ҩƷǰҪ�ȼ��װ�õ������ԡ�
��3��ʵ�����������ķ�Ӧԭ����п��ϡ���ᷴӦ��������п������������ʽΪZn+H2SO4=ZnSO4+H2����Cװ����Bװ����ȣ����ŵ��ǿ�����ʱ���Ʒ�Ӧ�ķ�����ֹͣ�����濪���á������ͣ������������������Լ�����������˼��������֣���
��4��ʵ�����ƶ�����̼�õ�Һ��ҩƷ��ϡ���ᣬb�Ǽ���Һ��ҩƷ�õģ���b��Ӧ����ϡ���Ũ��������ˮ�ԣ�������������������Ҫ��ȡ����Ķ�����̼����Ӧѡ��ʢ��Ũ�����Dװ�ã���Ϊ������̼���ܶȱȿ�������Eװ���ռ�CO2��Ӧ���������ſ�������Ҫ�����̳����������cͨ�롣
�ʴ�Ϊ����1�� a. �ƾ��� b. ����©��
��2��A�� ���װ�������ԣ� 2KMnO4 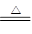 K2MnO4+ MnO2+O2�� �ֽⷴӦ
K2MnO4+ MnO2+O2�� �ֽⷴӦ
��3��Zn+H2SO4=ZnSO4+H2�� �û���Ӧ
������ʱ���Ʒ�Ӧ�ķ�����ֹͣ�����濪���á������ͣ��������������������˼��������֣�
��4��ϡ���ᣨ��ϡHCl�� CaCO3��2HCl=CaCl2��CO2����H2O
Ũ���ᣨ��ŨH2SO4�� c
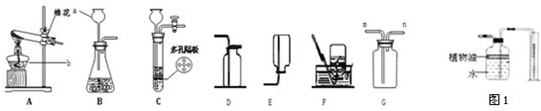
���㣺������ȡװ�õ�̽������������ȡװ�ã�������̼��ʵ�����Ʒ�����������ȡ�ͼ��顣
�������������ȡ�dz�����Ҫ�Ļ�ѧʵ��֮һ���������ص㣬���ǿ����ȵ㣬���ⷴӦԭ��������װ�õ�ѡ�����ݡ�װ�������Լ�鷽��������ļ��顢�������ռ��������˽�ʵ�鲽���ע�������ǽ����������Ĺؼ���

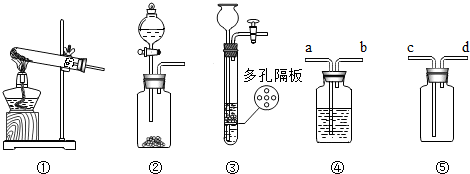
����ͼʾʵ��װ�ã��ش��������⡣
 |
��1��ijͬѧ��ʵ������Bװ�ã�����������ſ�״���壩��ȡ������̼����Ӧ�Ļ�ѧ����ʽΪ ��Bװ����Aװ����Ƚϣ��ŵ�Ϊ �����һ�㼴��������Cװ���ռ�CO2��������Ӧ�� ��ͨ�루�c����d������