��Ŀ����
18�� ��ͨ��ͭ����ͭ��п��ɣ�����һ���ͭ��Ʒ������Ϊ50.00g��ijѧϰС������һ��δ֪Ũ�ȵ�ϡ�������ⶨпͭ�Ͻ���ͭ������������ʵ������Ʒ�����ַ�Ӧ����Ʒ�������仯�����ͼ��ʾ��
��ͨ��ͭ����ͭ��п��ɣ�����һ���ͭ��Ʒ������Ϊ50.00g��ijѧϰС������һ��δ֪Ũ�ȵ�ϡ�������ⶨпͭ�Ͻ���ͭ������������ʵ������Ʒ�����ַ�Ӧ����Ʒ�������仯�����ͼ��ʾ����1����˻�ͭ��ͭ������������
��2��������������������
��3��������ͼ�е�tʱ����Ʒ��ϡ����ǡ����ȫ��Ӧ����ʱ��ü���ϡ������Һ���ΪV����Ϊ���ϵ���֪�����ϡ���������������������ȱ�ٵ�һ��������A��
A����Ӧǰ����ϡ������ܶȦ� B����Ӧ����ϡ���������
C��ʵ��ǰ��Ӧװ���п�������� V��D��ʵ���������������ܶȦѡ�
�����ԭ��������Zn��65 Cl��35.5 H��1������������һλС����
���� ��1�����ݻ�ͭ��ͭ�������ᷴӦ����ͼ1��֪��ͭ��ȫ��Ӧ����ʣ���������ͭ��������
��2�����ݷ�Ӧ�Ļ�ѧ����ʽ������ȫ��Ӧʱ����п���������������������������
��3�����������ܶȹ�ʽ�����ǡ�÷�Ӧʱ����������������н��
��� �⣺��1������ͭ���������ᷴӦ����ͼ1�ɵû�ͭ��ͭ������Ϊ33.75g��
��ͭ��Cu������������$\frac{33.75g}{50g}��100%$=67.5%��
��2����ͭ��п������=50.00g-33.75g=16.25g
����ȫ��Ӧ������H2������Ϊx��
Zn+2HCl�TZnCl2+H2��
65 2
16.25g x
$\frac{65}{16.25g}=\frac{2}{x}$
x=0.5g
�𣺣�1����ͭ��Cu����������67.5%����2����Ʒ��ȫ��Ӧ������H2������Ϊ0.5g��
��3�������ܶȹ�ʽ�����ǡ�÷�Ӧʱ����������������������÷�Ӧǰ����ϡ������ܶȦѡ�����ͼ�е�tʱ����Ʒ��ϡ����ǡ����ȫ��Ӧ����ʱ��ü���ϡ������Һ���ΪV���������ǡ�÷�Ӧʱ���������������п����������μӷ�Ӧ�Ȼ�����������������ϡ���������������������ѡ��A��
���� ���⿼���˸��ݻ�ѧ����ʽ������������������ļ��㣬���ڻ���֪ʶ�Ŀ��飮

 ��У����ϵ�д�
��У����ϵ�д�| A�� | ��ŨHCl���Լ�ƿ�������������Ũ������Լ�ƿҲ��������� | |
| B�� | ��������������ˮ����������Ҳ������ˮ | |
| C�� | ������ϡ���ᷴӦ����������ͭҲ����ϡ���ᷴӦ�������� | |
| D�� | ϡ������ʹ��ɫʯ����Һ���ɫ��ϡ����Ҳ��ʹ��ɫʯ����Һ���ɫ |
| A�� | �������ð�ˮ | B�� | �ü�ȩ��Һ���ݺ��� | ||
| C�� | ʳƷ������뵪���ӳ�ʳƷ������ | D�� | ʳ���߲��Բ���ά���� |
| A�� | 13.04% | B�� | 18.25% | C�� | 17.05% | D�� | ������ |
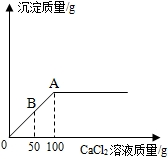 ��1��С����ⶨijƿ���ֱ��ʵ�NaOH��Һ�� Na2CO3��������������ȡ20g������Һ����������μ���1.11%��CaCl2��Һ��������������������CaCl2��Һ�����Ĺ�ϵ��ͼ��ʾ��
��1��С����ⶨijƿ���ֱ��ʵ�NaOH��Һ�� Na2CO3��������������ȡ20g������Һ����������μ���1.11%��CaCl2��Һ��������������������CaCl2��Һ�����Ĺ�ϵ��ͼ��ʾ��