��Ŀ����
��10�֣��������ĿҪ��ش���������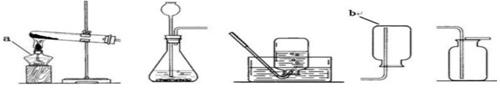
A B C D E
��1��ij����С�飬������ͼ�ṩ�IJ���װ�ã���ȡ���塣����ͼ����a������Ϊ_________��b��������__________����ȡ���ռ�������̼��ѡ��װ��Ϊ������ţ���ͬ��_______ ������װ�û�����ȡ���ռ�___________�������ø��������ȡ���ռ���������ѡ���װ��Ϊ__________���÷�Ӧ�Ļ�ѧ����ʽΪ______________��
��2��ʵ������п����ϡ������ȡ���ռ�������Ӧѡ�õ�װ��Ϊ������ţ�_______���÷�Ӧ�Ļ�ѧ����ʽΪ____________��
��1���ƾ��� ����ƿ B��E ���� A��E����A��C��
2KMnO4��K2MnO4+MnO2+O2��
��2��B��C����B��D�� Zn + H2SO4 = ZnSO4 + H2��
����

��ϰ��ϵ�д�
 ���������������Բ��������ϵ�д�
���������������Բ��������ϵ�д�
�����Ŀ
�±��ǵ������ڲ���Ԫ�ص���Ϣ���ϣ��������ĿҪ��ش��������⣮
��1���ڱ�����Ӧ�ĺ����ϣ���д��Ӧ�����ݣ�
| Ԫ������ | Ԫ�ط��� | ԭ�ӽṹʾ��ͼ | ��ߡ���ͻ��ϼ� | ��� |
| �� | Na |  | +1 | ���� |
| þ | Mg |  | +2 | ���� |
________ | Al |  | +3 | ���� |
| �� | Si |  | +4��-4 | ________ |
| �� | P |  | +5��-3 | �ǽ��� |
| �� | S | 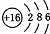 | +6��________ | �ǽ��� |
| �� | ________ |  | +7��-1 | �ǽ��� |

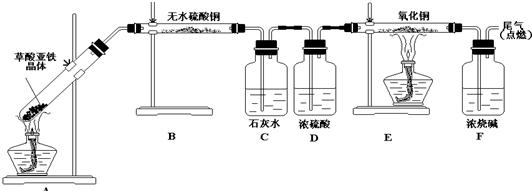
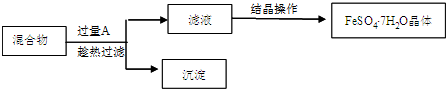







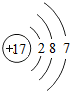
 �������ĿҪ��ش��������⣮
�������ĿҪ��ش��������⣮