��Ŀ����
2�� ˮ������֮Դ��Ҳ������������Դ��������ѧ���Ļ�ѧ֪ʶ���ش��������⣺
ˮ������֮Դ��Ҳ������������Դ��������ѧ���Ļ�ѧ֪ʶ���ش��������⣺��1����Щ��ׯ���ȡ�õ���ˮ���������ˮ��Ӳˮ������ˮ����ѡ�õ������Ƿ���ˮ��
��2����ͼ����ˮͨ��ֽ��ʾ��ͼ����ʵ������У��Թ�a�в�����������������д���÷�Ӧ�Ļ�ѧ����ʽ��2H2O$\frac{\underline{\;ͨ��\;}}{\;}$2H2��+O2����
��3��Ϊ��ȥˮ�еIJ��������ʣ�ijͬѧ������ͼ����ʾ�ļ���ˮ�������л���̿����Ҫ������������
��4��ijЩ�ط�������ˮ�к���������Ca��HCO3��2�ȿ������Σ���ˮʱ��Ca��HCO3��2�����ֽⷴӦ�����������Ե�CaCO3������CO2��ˮ����д��Ca��HCO3��2���ȷֽ�Ļ�ѧ����ʽCa��HCO3��2$\frac{\underline{\;\;��\;\;}}{\;}$CaCO3��+CO2��+H2O��
���� ��1������ˮ�ɼ�����ˮ��Ӳˮ��
��2�����ݵ��ˮʵ�������ͽ��۷����ش�
��3�����ݻ���̿�������Է����ش�
��3�����ݷ�Ӧԭ��д����Ӧ�ķ���ʽ��
��� �⣺��1���ӷ���ˮ��������ĭ�϶������ˮ��������ĭ���ٵ���Ӳˮ�����Է���ˮ�ܹ�������ˮ��Ӳˮ��
��2��ˮͨ��ֽ��������������������÷�Ӧ�Ļ�ѧ����ʽ�ǣ�2H2O$\frac{\underline{\;ͨ��\;}}{\;}$2H2��+O2������ʵ������У��Թ�b�в����������ǵ�Դ�������������������O2���Թ�a�в����������ǵ�Դ��������������϶���H2��
��3�����ڻ���̿�������ԣ��ɳ�ȥˮ�еIJ��������ʣ�ijͬѧ������ͼ�ҵļ���ˮ�������л���̿����Ҫ�������������ã�
��3��Ca��HCO3��2���ȷֽ����ˮ���Ļ�ѧ����ʽΪ��Ca��HCO3��2$\frac{\underline{\;\;��\;\;}}{\;}$CaCO3��+CO2��+H2O��
�ʴ�Ϊ��
��1������ˮ��
��2��������
��3��������
��4��Ca��HCO3��2$\frac{\underline{\;\;��\;\;}}{\;}$CaCO3��+CO2��+H2O��
���� ������Ҫ������ˮ����ɼ�������֪ʶ����ס���ˮ��ʵ��������ۼ�����̿���������ǽ����Ĺؼ���

| A�� | 4Fe+3O2$\frac{\underline{\;��ȼ\;}}{\;}$ 2Fe2O3 | B�� | 2KClO3 $\frac{\underline{\;MnO_2\;}}{\;}$2KCl+3O2�� | ||
| C�� | 4P+5O2$\frac{\underline{\;\;��\;\;}}{\;}$2P2O5 | D�� | C2H5OH+3O2$\frac{\underline{\;��ȼ\;}}{\;}$2CO2+3H2O |
| A�� | ���ʯ��ʯī��C60 | B�� | Һ�������������� | ||
| C�� | ʯ�͡���Ȼ�������� | D�� | ˮ�������ɱ� |
| A�� | 59.3% | B�� | 69.8% | C�� | 64.1% | D�� | 68.5% |

 ȼ�ϵķ�չ�ٽ������������IJ��Ͻ�����
ȼ�ϵķ�չ�ٽ������������IJ��Ͻ�����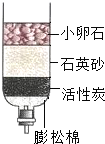 �ش��������⣺
�ش��������⣺ ijͬѧ��ʵ����������װ���Ʊ�������̽�����������ʣ�
ijͬѧ��ʵ����������װ���Ʊ�������̽�����������ʣ�