��Ŀ����
�������������ҹ��Ƽҵ����������°����˽��Ƽ����ư���������������Ƽ�������������ڱ��Ͱ���ˮ��(NH3��NaCl���ﵽ���͵���Һ)ͨ�� CO2��
���������ϣ�(1)�����Ƽ����Ҫ��Ӧ�� ��NaCl+NH3+CO2+H2O�TNaHCO3��+NH4Cl�� ��2NaHCO3 �� Na2CO3+H2O+CO2����
(2) NaHCO3��NH4Cl���ܽ��������ͼ��ʾ��
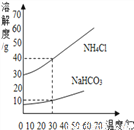
��˼��������(1)30��ʱ��NH4Cl������Һ����������������NaHCO3������Һ����������������ȣ�ǰ������ߵĹ�ϵ��_______(����ĸ)��
A������ B��С�� C������ D����ȷ��
(2)��Ӧ����������NaHCO3��NH4Cl��������Ϊ_______(���軯��)��
(3)����ˮ����CO2������NaHCO3��NH4Cl���Ƚᾧ������������NaHCO3��ԭ����________��
��ʵ��̽����ijС���Դ��κ�̼�����(NH4HCO3)Ϊԭ�ϣ��������������Ʊ������NH4Cl��
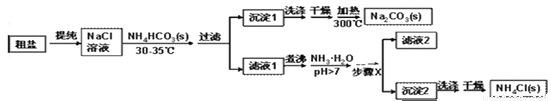
(1)�����г��˺�����ɳ�Ȳ��������ʣ�������������MgCl2�ȡ���Ҫ��ȥ�����е�MgCl2���ɼ��������NaOH��Һ�����ˣ�Ȼ������Һ�м����������ᡣд����������ʱ��Ӧ�Ļ�ѧ����ʽ_____________��
(2)����ʱ��������������______________ ��
(3)����1���泣����NH4+��Cl-�����ʣ�ϴ��ʱ���ѡ������______������Һ(����ĸ)��
A��NaCl B��NH4Cl C�� NH4HCO3 D��NaHCO3
(4)����X�����IJ�����________����ȴ�ᾧ���ˡ�
����չ���죩С�������ͼʵ��װ�òⶨij������Ʒ(����������NaCl)��̼���Ƶ�����������ȡһ��������Ʒ������ϡ���ᷴӦ��ͨ���ⶨ����CO2�������������Ʒ��̼���Ƶ�����������(��֪���³�ѹ��CO2���ܶ���1.977g/L)
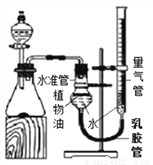
(1)�����Ʒ����Ϊ5.3g����ʵ����ѡ�õ������ܹ������ʵ���_________(����ĸ)��
A��500mL B��1000 mL C��1500 mL D��2000mL
(2) ��Ӧ��������ƿ����CO2�������ᵼ�²�õ�̼������������_________(����ĸ)��
A��ƫ�� B��ƫС C����Ӱ��
(3)���5.3g��Ʒ����Ԫ�ص���������Ϊ42%��ʵ���������ƿ����Һ���ɣ������ù�������Ϊ__________(�������1λС��)��
A 84:53.5 ��Ӧ���ɵ�̼�����Ƶ��������Ȼ�臨࣬����ͬ�¶���̼�����Ƶ��ܽ�ȱ��Ȼ��С NaOH+HCl=NaCl+H2O ���� D ����Ũ��(������) C C 5.7 g(��5.6 g) ���������� (2) ��˼��������(1) �ܽ����һ���¶��£�100g�ܼ���ﵽ����ʱ�����ܽ�����ʵ�������������Һ���ʵ���������=�ܽ�ȡ£��ܽ��+100g����100% �� 30��ʱ���Ȼ�淋���...ͬѧ�Ǵ�ɽ�ϲɼ���һ��ʯ��ʯ(��Ҫ�ɷ�Ϊ̼���),����ȡ80 g����Ʒ��������ʵ��(�������������չ����в������仯��̼��Ʒֽ�������ƺͶ�����̼),��÷�Ӧ����������Ƶ������뷴Ӧʱ��Ĺ�ϵ���±�:
��Ӧʱ��/s | t0 | t1 | t2 | t3 | t4 | t5 | t6 |
��Ӧ������ ����/g | 80 | 75 | 70 | 66 | 62 | 58 | 58 |
��ش���������:
(1)��ʯ��ʯ��ȫ��Ӧ��,����CO2������Ϊ_______________��
(2)���ʯ��ʯ�к�CaCO3����������(д���������)__________��
22 g 62.5%�� ����������1�����ڹ����������80g��Ϊ58g��������22g��˵�����ɵ����������Ϊ22g�� ��2���������ɵ����������Ϊ22g����˿��Ը������ɵ��������������������뷴Ӧ��̼��Ƶ������� �������� ����Ʒ�к�CaCO3������Ϊx CaCO3 CaO+CO2�� 100 44 x 22g x=50g ��Ʒ�е���������=��1...��ͼ��һ����״���������ʵ�����±����еķ���������ͼ����������
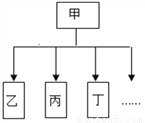
ѡ�� | �� | �� | �� | �� | �� |
A | Ӫ���� | ������ | ���� | ά���� | �� |
B | ���� | ���ʯ | ˮ�� | �ɱ� | �� |
C | ��Һ | ��� | ��ˮ | ��ˮ | �� |
D | ���� | ���� | �� | �ط� | �� |
A. A B. B C. C D. D
B ��������A��������Ҫ������Ӫ����Ϊ�������ʡ���֬�����ࡢά���ء�ˮ�����Σ���ȷ��B�����ʯ��ˮ������������������һ��Ԫ����ɵĴ�������ڵ��ʣ��ɱ���������̼�����ɲ�ͬԪ����ɵĴ�������ڻ��������C����ơ���ˮ ����ˮ���Ǿ�һ���ȶ��Ļ���������Һ����ȷ��D���������ʣ�ֻ���е����е�һ��Ԫ��ʱ������Ԫ���ǵ��ʣ�������Ԫ�����ʣ����м�Ԫ���Ǽطʡ�ͬʱ���е����е����ֻ�����...

 2����������Ԫ�صĻ��ϼ�Ϊ��
2����������Ԫ�صĻ��ϼ�Ϊ��