��Ŀ����
4��ij���������ð�������Ĵ����Ʒ�к��������Ȼ������ʣ����Ʒ��װ��ע����̼���ơ�96%��Ϊ�ⶨ�ò�Ʒ��̼���Ƶ�����������ͬѧ�ǽ���������ʵ�飺ȡ11.0�˴�����Ʒ�����ձ��У��Ƶ��ձ�����ʢ������Ʒ��������Ϊ158.0�ˣ��ٰ�100.0��ϡ����ƽ���ֳ��ķ����μ�����Ʒ�У���ʹ��ÿ�ζ��ܳ�ַ�Ӧ��ʵ�����ݼ�¼���£�| ��������Ĵ��� | ��һ�� | �ڶ��� | ������ | ���Ĵ� |
| �ձ�����ʢ���ʵ����������ˣ� | 181.2 | 204.4 | 228.6 | 253.6 |
��2���ò�Ʒ�е�̼�������������Ƿ����Ҫ�������ȷ��0.1%��
���� ��1�����������غ㶨�ɣ��ڻ�ѧ��Ӧ�У��μӷ�Ӧǰ�����ʵ������ܺ͵��ڷ�Ӧ�����ɸ����ʵ������ܺͣ��ʵ�һ�μ���ϡ�����ַ�Ӧ�����ɶ�����̼������=��Ӧǰ�ձ�����ʢ���ʵ������ܺ�-��Ӧ���ձ�����ʢ���ʵ������ܺͣ�
��2����ͼ�����ݿ�֪��ǰ���η�Ӧ��ÿ�������������������1.8g�����Ĵη�Ӧû���������ɣ��ʿ��жϵ����η�Ӧ����ȫ�����������������Ϊ158g+75g-228.6g=4.4g������̼������ϡ���ᷴӦ�Ļ�ѧ����ʽ�����ɵ������������г�����ʽ�����ɼ�������뷴Ӧ��Na2CO3��������Ȼ�����������������=$\frac{��������}{��Һ����}$��100%���㼴�ɣ�
��� �⣺��1����һ�εĽ���У�������̼������Ϊ158.0g+100g��4-181.2g=1.8g���ʴ�Ϊ��1.8g��
��2��ǰ���η�Ӧ��ÿ�������������������1.8g�����Ĵη�Ӧû���������ɣ��ʿ��жϵ����η�Ӧ����ȫ�����������������Ϊ158g+75g-228.6g=4.4g
����뷴Ӧ��Na2CO3������Ϊx��
Na2CO3+2HCl=2NaCl+CO2��+H2O
106 44
x 4.4g
��$\frac{106}{x}=\frac{44}{4.4g}$��
��֮�ã�x=10.6g��
��Ʒ��̼���Ƶ���������Ϊ��$\frac{10.6g}{11g}$��100%��96.4%��96%��
�ʸò�Ʒ��̼���Ƶ����������ϸ�
���� ������Ҫ����ѧ�����û�ѧ����ʽ�ۺϷ����ͽ��ʵ�������������������ѧ�����������˼ά��ȣ�ǿ����ѧ������֪ʶ��������

 Ӧ������ҵ��ϵ�д�
Ӧ������ҵ��ϵ�д�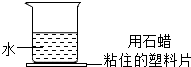 С÷ͬѧ����ͼ��ʾ���ձ��м���һ�����ʣ������������Ƭ�ϵ�ʯ���ڻ���������������ǣ�������
С÷ͬѧ����ͼ��ʾ���ձ��м���һ�����ʣ������������Ƭ�ϵ�ʯ���ڻ���������������ǣ�������| A�� | ʳ�ι��� | B�� | �������ƹ��� | C�� | ������ | D�� | ����粒��� |
| A�� | ����һ�ֽ���Ԫ�أ��������� | B�� | �ֵĻ�ѧ���ʲ����� | ||
| C�� | ����ɫ���������ֵ��������� | D�� | ϡ������һ�ֻ���� |
| A�� |  �ò������ò��� | B�� |  þ��ȼ�� | ||
| C�� | 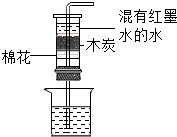 ľ̿��ˮ | D�� |  ˮ���� |
��2H ��2NH3 ��SO2 ��Mg2+ ��2OH- ��H2O��
| A�� | ��ʾ���ӵĸ������Ǣܢ� | B�� | ��ʾ����������������Ǣܢ� | ||
| C�� | ��ʾ���Ӹ������Ǣ٢� | D�� | ��ʾ������ԭ�Ӹ������Ǣۢ� |
| A�� | Mg��OH��2+2HCl�TMgCl2+2H2O | B�� | 2HCl+Na2SO4�T2NaCl+H2SO4 | ||
| C�� | CaCl2+Na2CO3�TCaCO3��+2NaCl | D�� | Fe2O3+6HCl�T2FeCl3+3H2O |