��Ŀ����
��ѧ������ѧϰ��ѧ����Ҫ���ߡ�
��1�����л�ѧ���Ŷ�������д���������Ը�����
3��������(3N)________��1��������(Na1+)________������þ(MgO2)________��
��2����Ҫ��д�����л�ѧ���š�
�ؿ��к������Ľ���Ԫ��________�����ֶ�����̼��ѧ���ʵ���С��________��
3N2Na+MgOAlCO2 �������� (1) ���ʵĻ�ѧʽ����д����Ԫ�ط������½DZ���һ��������ԭ�ӵĸ�������ѧʽǰ�ߵ����ֱ�ʾ���ӵĸ���3�������ӣ�3N2�����ӷ�����д��Ԫ�أ�������������Ͻǵ����ֱ�ʾһ�����Ӵ���ɵ���ֵ�������������������ң������Ϊ1ʱ��1��д��1��������Na+��������Ļ�ѧʽ��д�������۵�Ԫ�������д����ߣ��Ը��۵�д���ұߣ����ϼ۵ľ���ֵ����Լ�����û�ѧʽ...��������ͼ���е���Ϣ��ѧϰ��ѧ��һ����Ҫ������
��1���±����Ȼ��ƺ�̼�����ڲ�ͬ�¶�ʱ���ܽ�ȣ����ݴ˱��ش�
�¶�/�� | 10 | 20 | 30 | 40 | |
�ܽ��/g | �Ȼ��� | 35.8 | 36.0 | 36.3 | 36.6 |
̼���� | 12.2 | 21.8 | 39.7 | 53.2 |
��40��ʱ���Ȼ��Ƶ��ܽ��Ϊ________ g��
��̼���Ƶ��ܽ�����¶ȵ����߶�________�������С��������20��ʱ����100g��ˮ����30g̼�����У���ֽ����õ�����________ ���������Һ����������Һ��������������Һ���µ�30�棬����Һ��������������Ϊ________ ����������ȷ��0.1%����
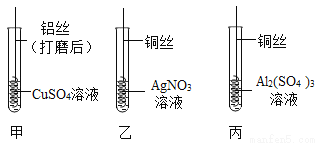
��2����������ϡ�������һ����������ͭ������У�д�����з�Ӧ�Ļ�ѧ����ʽ________����ͼ��ʵ��������������ʣ�����������淴Ӧʱ��ı仯��ϵ�����б�ʾ��ȷ����________ �����ţ���




 2Fe203
2Fe203