��Ŀ����
18�������2014���п���ѧʵ�鿼���У�������60g5%���Ȼ�����Һ��������̼��ʵ������ȡ�����кͷ�Ӧ����������10mL��ʳ��ˮ���ĸ���ǩ����ѧ����ǩȷ��һ��������п��죮��1����ͬѧ��ǩ��ʵ���ң����ֱ���ʵ��������������������ҩƷ��
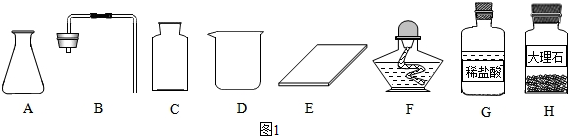
������������D������Ϊ�ձ���F������Ϊ�ƾ��ƣ�
�ڼ�ͬѧ�鵽�Ŀ�ǩӦ���Ǣ�����ĸ��ţ���
����ȡһƿ�����壬Ӧѡ�õ�������ABCE������ĸ��ţ���
�������������巢��װ����ʵ���һ�������ȡ�������������壬����д����ȡ������Ļ�ѧ����ʽ2H2O2$\frac{\underline{\;MnO_2\;}}{\;}$2H2O+O2�����÷�Ӧ����Ϊ�ֽⷴӦ��
��2����ͬѧ�鵽�Ŀ�ǩ�Ǣ�����ʵ�������ͼ2��ʾ����ش�
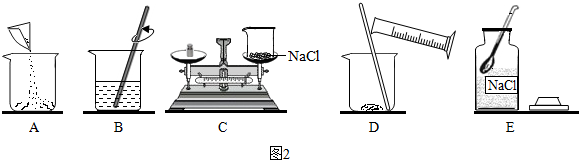
��ͼʾ�����У���һ���Ǵ���ģ���һ���������C����д��ĸ��ţ���
������������ȷ��˳����ECADB����д��ĸ��ţ���
�۰�����ȷ�IJ��������ƺõ���Һװ���Լ�ƿʱ�����������˼��Σ����ʹ��Һ�����ʵ������������䣨�ƫ��ƫС�����䡱����
��3����ͬѧ�鵽�Ŀ�ǩ�Ǣ������ڽ���ʵ��ʱ�������õ�����Һ���ǣ����������õ�����Һ�����˵ڶ��ι��ˣ����������Һ��Ȼ���ǣ����������ͬѧ����һ�²����������ԭ����Һ������ֽ��Ե��д��һ�㼴�ɣ���
���� ��1���ٸ���ʵ���ҳ������������ƺ�������ָ���������ý��з�����
�ڸ������������ƺ�ҩƷ����ѡ��
�۸��ݹ����Һ���ڳ����µķ�Ӧѡ����װ�úͶ�����̼���ܶȺ��ܽ���ѡ���ռ���������Ӧ��������
�ܸ��ݷ���װ�����ڹ�Һ������װ�ã�������ȡ�������з�����
��2���ٸ�����ƽ��������Ӧ��������������з�����
�ڸ�������һ����������������Һ����ȷ����������з�����
�۸��ݽ����ƺõ���Һװ���Լ�ƿʱ�����������˼��Σ�����Ӱ����Һ�����������з�����
��3�����ݹ��˵���ȷ����������з�����
��� �⣺��1����ͨ������������ָ���������ÿ�֪��D���ձ���F�Ǿƾ��ƣ�
�ڸ��ݸ�����ҩƷϡ����ʹ���ʯ����������ͬѧ�鵽�Ŀ�ǩӦ���ǣ�������̼����ȡ����ѡ����
����ȡ������̼��ҩƷ��ʯ��ʯ��ϡ���ᣬ����װ���ǹ����Һ���ڳ����µķ�Ӧ����Ҫ�������У�AB����Ϊ������̼������ˮ���ܶȱȿ��������Բ��������ſ������ռ�����Ҫ�������У�CE��
�ܷ���װ�����ڹ�Һ�����͵�װ�ã���˿�����ʵ�����ù���������Һ�Ͷ���������ȡ���������ж��������Ǵ�������ѧ����ʽΪ��2H2O2$\frac{\underline{\;MnO_2\;}}{\;}$2H2O+O2�����÷�Ӧ����ֽⷴӦһ�������������ڷֽⷴӦ��
��2������ƽ��������Ӧ�����������룬���������C��
������һ����������������Һ����ȷ���������Ǽ��㡢�������ܽ⣬���Բ�����ȷ��˳����ECADB��
�۽����ƺõ���Һװ���Լ�ƿʱ�����������˼��Σ�����Ӱ����Һ��������������ʹ��Һ�����ʵ������������䣻
��3���ڹ��˹����У������ֽ����Һ�������ֽ��Ե������ʹ��Һ���еIJ��������������ձ����Ӷ�ʹ����Һ���ǣ��н���Һ���ձ����ɾ�ʱҲʹ����Һ���ǣ����Ի��ǵ�ԭ���У�����ֽ���𣬢��������ɾ�����©����Һ�泬����ֽ��Ե��
�ʴ�Ϊ������1�����ձ����ƾ��ƣ��ڢ�ABCE����2H2O2$\frac{\underline{\;MnO_2\;}}{\;}$2H2O+O2�����ֽⷴӦ��
��2����C����ECADB���۲��䣻��3����Һ������ֽ��Ե��
���� ���⿼���˳����������ȡװ�á��ռ������ȵ�ѡ���й���Һ�ļ��㣬���ڴ˻����Ͻ�����չ���ؼ�����ȷ����װ�á��ռ�����ѡ������ݣ�

| A�� | -2 | B�� | 0 | C�� | +2 | D�� | +4 |
| A�� |  ��ȡҺ�� | B�� |  ��������ζ | C�� |  �����鴿 | D�� |  ϡ��Ũ���� |
| A�� | ��ͭ��ͭп�Ͻ𣩱�ͭ��Ӳ��С | |
| B�� | ���ֽ����У�����ͭ�ĵ��������� | |
| C�� | ������ͭ�������ǰ��ս���������������ǿ��˳�����е� | |
| D�� | ���ֽ����У�ֻ�����ֽ����ܺ��ᷢ���û���Ӧ�������� |
��Co+2HCl�TCoCl2+H2��
��CoCl2+2NaOH�TCo��OH��2+2NaCl
���ж�������Τ��˵���в���ȷ���ǣ�������
| A�� | �������ܵĻ�ѧ�����ȶ� | B�� | �������ܿ��Ը����Ȼ������� | ||
| C�� | ��Ϊ�û���Ӧ����Ϊ���ֽⷴӦ | D�� | Co��OH��2��CoԪ�صĻ��ϼ�Ϊ+4 |
| A�� | 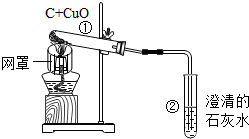 ��ͼ�Թܢ��к�ɫ��ĩ��ɺ�ɫ���Թܢ��еij���ʯ��ˮ����ǣ�˵��̼���п�ȼ�� | |
| B�� | 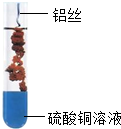 ��ͼ��˿���渲��һ���ɫ���ʣ���Һ����ɫ�����ɫ��˵�����������ǿ��ͭ | |
| C�� |  ��ͼ�ձ��еĺ�ɫ��Һ��dz�������ɫ��˵���������������Ʒ�Ӧ����Һ������ | |
| D�� |  ��ͼ��˿�������о���ȼ�ջ����������ɺ�ɫ�����������壬˵���������������� |
 ��ͼ�dz��л�ѧ�м��ֳ������ʼ�ķ�Ӧ��ת����ϵ��A���ʾ��л�ԭ�ԣ�B��һ�ֳ��������壬E��ˮ��Һ����ɫ����ͼ�С�-����ʾ���˵������ܷ�����Ӧ����������ʾһ�����ʿ�ת��Ϊ��һ�����ʣ�
��ͼ�dz��л�ѧ�м��ֳ������ʼ�ķ�Ӧ��ת����ϵ��A���ʾ��л�ԭ�ԣ�B��һ�ֳ��������壬E��ˮ��Һ����ɫ����ͼ�С�-����ʾ���˵������ܷ�����Ӧ����������ʾһ�����ʿ�ת��Ϊ��һ�����ʣ�