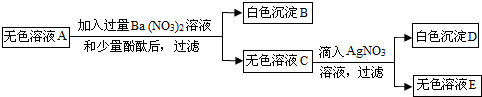
��Ŀ����
16���ס�����ͬѧ�ֱ�����NaOH��Һ��ϡ���Ტ�������ߵ��кͷ�Ӧʵ�飮��1����ͬѧ����80g��������Ϊ10%��NaOH��Һ�������������̷����ձ�����������һ����������������룬���������������ƣ�ֱ����ƽƽ�⣻
��2����ͬѧ��98%��Ũ��������20%��ϡ���ᣬ������Ͳ��ȡŨ����ʱ���Ӷ���������������Һ����������������ƫ��ѡ�ƫ����ƫС�����䡱��
��3����Ũ����ϡ�͵ķ����ǽ�Ũ�������������ձ��ڻ���ע��ˮ�У����ò��������Ͻ��裻
��4�����к�����NaOH��Һ����ȡ20%��ϡ��������Ϊ49g��
���� ��1�����ݳ�������������Ҫ���ձ��г�������ֹ��Ⱦ���̽��з�����
��2���������Ӷ�����õ�Ũ�������ƫ����������Һ����������������ƫ����з�����
��3������ϡ��Ũ�������ȷ�������з�����
��4�����ݻ�ѧ����ʽ�����е����ݽ��м��㣮
��� �⣺��1����������������Ҫ���ձ��г�������ֹ��Ⱦ���̣�
��2�����Ӷ�����õ�Ũ�������ƫ����������Һ����������������ƫ��
��3��ϡ��Ũ����ʱ����Ũ�������������ձ��ڻ���ע��ˮ�У����ò��������Ͻ��裻
��4����ȡ20%��ϡ��������Ϊx
2NaOH+H2SO4=Na2SO4+2H2O
80 98
80g��10% x��20%
$\frac{80}{80g��10%}$=$\frac{98}{x��20%}$
x=49g
�ʴ�Ϊ����1���ձ���
��2��ƫ��
��3����Ũ�������������ձ��ڻ���ע��ˮ�У�
��4��49��
���� ������Ҫ��������Һ�����йص�֪ʶ���ѶȲ�����Ҫ��ƽʱ��ǿѵ��������ɣ�

��ϰ��ϵ�д�
�����Ŀ
6�������йء��غ㡱�������У���ȷ���ǣ�������
| A�� | þ���ڿ�����ȼ�պ��������������ԭþ����������� | |
| B�� | �ڷ�Ӧ3Cu+8HNO3�T3Cu��N03��2+2X��+4H20�е�X�Ļ�ѧʽ��NO2 | |
| C�� | ����24.5gKClO3��4gMnO2�Ļ�����Ӧһ��ʱ���ʣ�����24.9g����O2������Ϊ3.6g | |
| D�� | ij��������������ȼ�պ�����CO2��SO2��CO����������һ������C��S��OԪ�� |
4�������й�CO��CO2�ıȽ��У���ȷ���ǣ�������
| A�� | ���ӹ�����ͬ | B�� | ���Ԫ��������ͬ | ||
| C�� | ����������ͬ | D�� | ��ѧ������ͬ |
1�����������У������л�����ǣ�������
| A�� | CO2 | B�� | KClO3 | C�� | CaCO3 | D�� | CH3COOH |
6��������������;�У������������������ʵ��ǣ�������
| A�� | ұ������ | B�� | ���ӽ��� | ||
| C�� | ��Ϊ��������ȼ�� | D�� | ���̽������ |
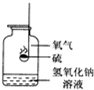 �����ͼ��ʾ��ȼ�յ�ʵ�飬�ش��й����⣮
�����ͼ��ʾ��ȼ�յ�ʵ�飬�ش��й����⣮