��Ŀ����
20�����ⶨij�Ȼ�泥�ֻ����������ʣ���Ʒ���Ȼ�淋������������ס�����λͬѧ����ͼ��ʾ��ʵ�鲽�����ʵ�飺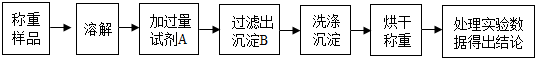
��1��ʵ��ʱ����ͬѧʹ����������Һ���Լ�A����ͬѧѡ���Ȼ�����Һ���Լ�A��������ѡ����Լ�A���������Ǽ�ͬѧ��
��2�����¶���ָ��ȷʹ���Լ�Aʱ���е�ʵ�飺
��Ϊ���г������ܽ⡢��Ӧ�����ˡ�ϴ�ӳ����ĸ����������������������������ƽ�����롢�ձ���������������̨����������ͷ�ιܡ���Ͳ�����л�ȱ�ٵ�һ���ر��IJ��������Dz�������
�ڼ����жϳ���B�Ƿ�ϴ���ķ�����ϴ�ӳ������õ�Һ���м��뼸����������Һ�����ް�ɫ������ϴ����
�����������ó���δ��ϴ�Ӽ���ɳ��أ������Ʒ���Ȼ�淋�����������ƫ�ͣ��ƫ�ߡ�����ƫ�͡��������䡱����
���� ��1�����������Ӻ���������ӵ����������
��2���ٸ��ݸ������������Ҫ�������������
�ڸ��������ӵļ��鷽�������н��
�۸��ݲ����ij�������������ϻ�ѧ����ʽ�����������淋�������������Ȼ�淋�����������
���������δ��ϴ�ӣ����ڳ������渽���������ƣ�����ֱ�Ӻ�ɵ��³������������ӣ����Ծݴ˽��
��� �⣺��1�������ӿ��Ժ����������γ����ڵ����ʣ���������Ҳ���Ժ������ӷ�Ӧ���ɰ�ɫ���������Ի����ʵ������ĵó�������ͬѧ����ѡ����Լ����ԣ�
��2���ٸ��������еIJ�������֪�������ܽ⡢���˺������ж�Ҫ�õ������������Կ���֪����������������ȱ�ٲ�������
��������û��ϴ��������ϴ��Һ�������������ӣ������ӿ��Ժ����������ɰ�ɫ���������Կ���������ϴ��Һ�е�����������Һ�����û�а�ɫ����������˵��ϴ�ɾ��ˣ������ûϴ�ɾ���
���������δ��ϴ�ӣ����ڳ������渽���������ƣ�����ֱ�Ӻ�ɵ��³������������ӣ�ʹ����ó�������淋��������ӣ��Ӷ�ʹ��������Ȼ�淋��������٣����Լ���õ����Ȼ�淋���������ƫ�ͣ�
�𰸣���1���ף���2���ٲ�����������ϴ�ӳ������õ�Һ���м��뼸����������Һ�����ް�ɫ������ϴ������ƫ�ͣ�
���� �������������Ӻ���������ӵļ���������������д��ѧ����ʽ���������ǽ��л�ѧ��������ݣ�Ҳ�����ǽ��б������յ�֪ʶ�㣮

| A�� | 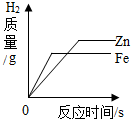 �����������״��ͬ��пƬ����Ƭ�ֱ���������ĵ�����������ϡ���� | |
| B�� | 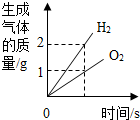 ���ˮ | |
| C�� | 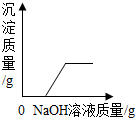 ���Ȼ���������Ļ����Һ�м������������������Һ | |
| D�� | 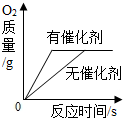 ��������ͬ������ͬ���������Ĺ���������Һ��ȡ���� |
| A�� | ���ʵ����Ա������� | B�� | ����֭�Լ��� | ||
| C�� | ��ľ��ˮ�ļ��Խ�ǿ | D�� | �ܲ������� |
| A�� | 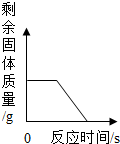 ��ʾһ������ľ̿��ԭ����ͭ��ʣ����������뷴Ӧʱ��Ĺ�ϵ | |
| B�� | 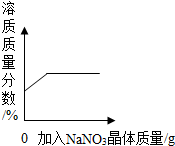 ��ʾt��ʱ����������Һ�м��������ƾ��壬��������������������Ĺ�ϵ | |
| C�� | 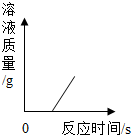 ��ʾһ������ϡ������п����Ӧ����Һ�������뷴Ӧʱ��Ĺ�ϵ | |
| D�� | 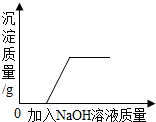 ��ʾ��������Ȼ�ͭ�����Һ�м����������ƣ�����������������Ĺ�ϵ |
| A�� | п������һ�ֱ������Ԫ�أ�ȱ�ٻ����������ٻ����������� | |
| B�� | �������ס���ʳ�����Ҫ�ɷ֣���������̼���⡢������Ԫ����ɵ� | |
| C�� | ÿ����֬����������ȫ�������ų��������������һ�����ϣ�����Ҫ�Ĺ������� | |
| D�� | ����ÿ��Լ��3-5g��ʳ�������������������Ҫ |
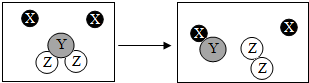 ����ѧ�仯�������磬�����ڽ�һ����ʶ��ѧ�仯����ͼ��ij��ѧ�仯����ʾ��ͼ��X��Y��Z�ֱ��ʾ��ͬ����ԭ�ӣ���ͼ�ش����⣺
����ѧ�仯�������磬�����ڽ�һ����ʶ��ѧ�仯����ͼ��ij��ѧ�仯����ʾ��ͼ��X��Y��Z�ֱ��ʾ��ͬ����ԭ�ӣ���ͼ�ش����⣺ ��Ȼ����һ����Ҫ�ġ���ʯ�ͻ�����Դ����ˮ������һ���ڸ�ѹ��������������ˮ����Ȼ���γɵı�̬��--��ȼ������Ҫ�ֲ��ں���½�ض������������У�����̽������Ȼ��ˮ����洢������ش��������⣺
��Ȼ����һ����Ҫ�ġ���ʯ�ͻ�����Դ����ˮ������һ���ڸ�ѹ��������������ˮ����Ȼ���γɵı�̬��--��ȼ������Ҫ�ֲ��ں���½�ض������������У�����̽������Ȼ��ˮ����洢������ش��������⣺ ��ͼΪ�г��ϳ��۵�һ�ּӵ�����ε����˵�������һ����ÿ������4g�ӵ�����Σ���ÿ��������Ԫ�ص�����Լ1.1g����������ȷ��0.1����
��ͼΪ�г��ϳ��۵�һ�ּӵ�����ε����˵�������һ����ÿ������4g�ӵ�����Σ���ÿ��������Ԫ�ص�����Լ1.1g����������ȷ��0.1����