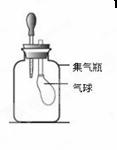
��Ŀ����
������������ʵ��װ��ͼ�ش����⡣
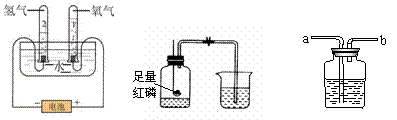 |
A�����ˮ B���ⶨ�������������� C��ϴ��ƿ
��1��Aʵ���У��Թ�2���Թ�1������������������ ��
| |
��2��Bʵ��˵������Լռ��������� �����в��������� ������ţ���
�ټ��װ��������  ��ʵ��ǰ�н�ֹˮ�� �ۺ���Ϩ���������ֹˮ��
��ʵ��ǰ�н�ֹˮ�� �ۺ���Ϩ���������ֹˮ��
��3������Cװ�ø���H2��ϴ��ƿ�е�Һ���Լ������� ��ҽԺ���ô�װ�����۲첡�˵��������������
���� ��a����b����Ӧ���Ӳ������������ܽ��ܡ�
��a����b����Ӧ���Ӳ������������ܽ��ܡ�
��1��2:1��ˮ�����⡢��Ԫ����ɵġ� ��2��1/5�� �� ��3��Ũ���b

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д���ij������Ʒ�У�����������������Ϊ10%������ͭ��Һ160 g��ǡ����ȫ��Ӧ����Ʒ�����ʲ�����ˮҲ��������ͭ��Ӧ��������ͼ�ṩ�����ݼ��㡣
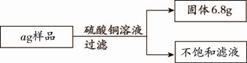 |
��1��a����ֵ��
��2��������Һ�м���40 .8 gˮ����������Һ�����ʵ�����������
.8 gˮ����������Һ�����ʵ�����������
Ϊ�˲ⶨij��Ʒ��̼�ᱵ��������������������ʵ�顣ȡһ��������Ʒ��400g������������Ϊ10%��ϡ��������ձ��С��ڻ�ѧ��Ӧ�����ж��ձ������е�ʣ�����������γ�������¼���±��������跴Ӧ���ٽ��У����ʲ�����ˮ��Ҳ����ϡ���ᷴӦ���ձ�������Ϊ25.4g��
| ��Ӧʱ�� | t1 | t2 | t3 | t4 | t5 |
| �ձ���ҩƷ����/g | 516.6 | 507.8 | m | 503.4 | 503.4 |
����㣺����Ӧ����ʽΪ��BaCO3 + 2HCl = BaCl2 + H2O + CO2����
��1�����еġ�m��Ϊ��
��2���ò�Ʒ��̼�ᱵ������������
��3������Ʒ��̼�ᱵ��ϡ����ǡ����ȫ��Ӧʱ��������Һ�����ʵ�����������