��Ŀ����
����Ԫ�ؿ�������������ʣ����ݱ��е�Ԫ�ػش��������⣮
| Ԫ������ | �� | ̼ | �� | �� | �� |
| Ԫ�ط��� | H | C | O | Cl | Ca |
��2��ij���嵥������ȫȼ�պͲ���ȫȼ��ʱ�����ɲ�ͬ�����壮�ù�����ȫȼ��ʱ��Ӧ�Ļ�ѧ����ʽΪ ����ҵ���øù����ˮ�����ڸ����������Ƶ�ȼ��ˮú������Ҫ�ɷ���CO��H2�����÷�Ӧ�Ļ�ѧ����ʽΪ ��
��3����ͼ��ʾʵ���У�����Һ©���Ļ�����ʹ����ˮ������ƿ�У�ʵ���N��Һ����ǣ���N��Һ�е������� ���Թܢ��е������� ���������������ԭ���� ��
���𰸡���������1��������������������Դ�������
��2��ij���嵥������ȫȼ�պͲ���ȫȼ��ʱ�����ɲ�ͬ�����壮���֪�ù��嵥����̼��������ɵ���Ϣ��дˮú����ȡ�ķ�Ӧ����ʽ��
��3����ʵ���N��Һ�����˵���ڵڢڸ��Թ��в����˶�����̼���壬N��Һ��ʯ��ˮ����һ��װ�õ��������뷨ʹ�Թܢ���Һ�������Թܣ�����ѹǿ������ʵ�ָ�Ŀ�꣬�ٽ�һ���ش����⣮
����⣺��1��������ѧ��֪��������������Դ������Ҫԭ��������ȼ�պ������ˮ���䷽��ʽΪ��2H2+O2 2H2O��
2H2O��
��2������ij���嵥������ȫȼ�պͲ���ȫȼ��ʱ�����ɲ�ͬ�����壮���Ըù��嵥��Ϊ̼�ĵ��ʣ���ȫȼ��ʱ�ķ���ʽΪC+O2 CO2���������������֪��ȡˮú���ķ�Ӧ����ˮ��̼����������һ����̼��ˮ�����Ը÷�Ӧ�ķ���ʽΪ��C+H2O
CO2���������������֪��ȡˮú���ķ�Ӧ����ˮ��̼����������һ����̼��ˮ�����Ը÷�Ӧ�ķ���ʽΪ��C+H2O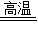 CO+H2��
CO+H2��
��3����ʵ���N��Һ�����˵���ڵڢڸ��Թ��в����˶�����̼���壬N��Һ��ʯ��ˮ����һ��װ�õ��������뷨ʹ�Թܢ���Һ�������Թܣ�����ѹǿ������ʵ�ָ�Ŀ�ֻ꣬Ҫ��������ˮ���ȾͿ��Դﵽ��Ŀ�꣬������������ˮ���ȵ��У��������ƹ�������ˮ���ȣ���ʯ������ˮ���ȣ����ڷ���ѹǿ�����Թܢ���Һ��ѹ���Թܢڣ������Թܢ����Ƿ�ĩ�������Թܢ���Һ�������ᣬ�Թܢ��з�ĩ��̼���Σ���������Ϊ�Թܢ���Һ�������Թܢ��У��Թܢ��з�ĩ���ٲ������ݲ���������ʵ���N��Һ������Ƕ�����̼���������Ʒ�Ӧ����̼��Ƴ�����ˮ��
�ʴ�Ϊ����1��2H2+O2 2H2O����2��C+O2
2H2O����2��C+O2 CO2��C+H2O
CO2��C+H2O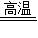 CO+H2��
CO+H2��
��3��Ca��OH��2���Թܢ���Һ�������Թܢ��У��Թܢ��й����ĩ���ٲ������ݲ�����CaO��ˮ��Ӧ���ȣ���ƿ���������ͣ�ѹǿ�����ʹ�Թܢ���ѹǿ��������ѹ���Թܢ��У���CaCO3������Ӧ����������CO2��
������������Ҫ����ѧ��������ѧ��ѧ֪ʶ�ۺϷ����ͽ��ʵ�������������������ѧ�����������˼ά��ȣ�ǿ����ѧ������֪ʶ��������
��2��ij���嵥������ȫȼ�պͲ���ȫȼ��ʱ�����ɲ�ͬ�����壮���֪�ù��嵥����̼��������ɵ���Ϣ��дˮú����ȡ�ķ�Ӧ����ʽ��
��3����ʵ���N��Һ�����˵���ڵڢڸ��Թ��в����˶�����̼���壬N��Һ��ʯ��ˮ����һ��װ�õ��������뷨ʹ�Թܢ���Һ�������Թܣ�����ѹǿ������ʵ�ָ�Ŀ�꣬�ٽ�һ���ش����⣮
����⣺��1��������ѧ��֪��������������Դ������Ҫԭ��������ȼ�պ������ˮ���䷽��ʽΪ��2H2+O2
 2H2O��
2H2O����2������ij���嵥������ȫȼ�պͲ���ȫȼ��ʱ�����ɲ�ͬ�����壮���Ըù��嵥��Ϊ̼�ĵ��ʣ���ȫȼ��ʱ�ķ���ʽΪC+O2
 CO2���������������֪��ȡˮú���ķ�Ӧ����ˮ��̼����������һ����̼��ˮ�����Ը÷�Ӧ�ķ���ʽΪ��C+H2O
CO2���������������֪��ȡˮú���ķ�Ӧ����ˮ��̼����������һ����̼��ˮ�����Ը÷�Ӧ�ķ���ʽΪ��C+H2O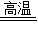 CO+H2��
CO+H2����3����ʵ���N��Һ�����˵���ڵڢڸ��Թ��в����˶�����̼���壬N��Һ��ʯ��ˮ����һ��װ�õ��������뷨ʹ�Թܢ���Һ�������Թܣ�����ѹǿ������ʵ�ָ�Ŀ�ֻ꣬Ҫ��������ˮ���ȾͿ��Դﵽ��Ŀ�꣬������������ˮ���ȵ��У��������ƹ�������ˮ���ȣ���ʯ������ˮ���ȣ����ڷ���ѹǿ�����Թܢ���Һ��ѹ���Թܢڣ������Թܢ����Ƿ�ĩ�������Թܢ���Һ�������ᣬ�Թܢ��з�ĩ��̼���Σ���������Ϊ�Թܢ���Һ�������Թܢ��У��Թܢ��з�ĩ���ٲ������ݲ���������ʵ���N��Һ������Ƕ�����̼���������Ʒ�Ӧ����̼��Ƴ�����ˮ��
�ʴ�Ϊ����1��2H2+O2
 2H2O����2��C+O2
2H2O����2��C+O2 CO2��C+H2O
CO2��C+H2O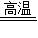 CO+H2��
CO+H2����3��Ca��OH��2���Թܢ���Һ�������Թܢ��У��Թܢ��й����ĩ���ٲ������ݲ�����CaO��ˮ��Ӧ���ȣ���ƿ���������ͣ�ѹǿ�����ʹ�Թܢ���ѹǿ��������ѹ���Թܢ��У���CaCO3������Ӧ����������CO2��
������������Ҫ����ѧ��������ѧ��ѧ֪ʶ�ۺϷ����ͽ��ʵ�������������������ѧ�����������˼ά��ȣ�ǿ����ѧ������֪ʶ��������

��ϰ��ϵ�д�
�����Ŀ
 ��ʾ̼ԭ�ӣ�
��ʾ̼ԭ�ӣ� ��ʾ��ԭ�ӣ�
��ʾ��ԭ�ӣ� ��ʾ��ԭ�ӣ�
��ʾ��ԭ�ӣ�
