��Ŀ����
�����ǵ���Ҫ�ɷ���̼���(�����ɷֲ�����ˮҲ�����ᷴӦ)����ѧ��ȤС��Ϊ�˲� ����������̼��Ƶĺ�����������ʵ�飺
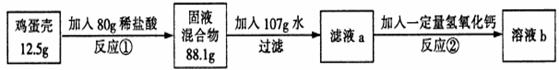
��Ӧ�ٽ�������������պ÷�Ӧ��һ�룬��Ӧ��ǡ����ȫ��Ӧ����ش��������⣺
��1����Ӧ�ٵĻ�ѧ����ʽΪ___________________________��
��2��������֪�����г����̼�������(X)�ı���ʽ______________��
��3���ü�������̼��Ƶ���������Ϊ_______��
��4�������������Ƶ�����Ϊ_______��
��5����Һb�����ʵ���������Ϊ_______��
��6����36.5����Ũ��������80g����ϡ�������ˮ������Ϊ_______��
��1��CaCO3+2HCl====CaCl2+H2O+CO2��(1��)����2��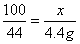 ����
����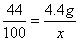 ��
��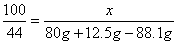 ��
��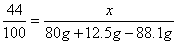 ������3��80��(1��)����4��7.4g(1��)����5��11.1��(1��)����6��40g(1��)
������3��80��(1��)����4��7.4g(1��)����5��11.1��(1��)����6��40g(1��)
��������
�����������1�������Ǻ���̼��ƣ��������ᷴӦ�����Ȼ��ơ�ˮ�Ͷ�����̼��CaCO3+2HCl==CaCl2+H2O+CO2������2���������غ㶨�ɿɵã���Ӧ�����ɵĶ�����̼������Ϊ��12.5g+80g-88.1g=4.4g,�ٽ�ϻ�ѧ����ʽ�����г����̼�������(X)�ı���ʽ��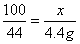 ����3���ɣ�2������ɵ�̼��Ƶ�X����Ϊ10g���ʸü�������̼��Ƶ���������Ϊ
����3���ɣ�2������ɵ�̼��Ƶ�X����Ϊ10g���ʸü�������̼��Ƶ���������Ϊ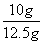 ��100%=80%����4���ɣ�2����ѧ����ʽ����ɵ÷�Ӧ���Ȼ�������Ϊ7.3g����Ϊ��Ӧ�ٽ�������������պ÷�Ӧ��һ�룬����Һa���Ȼ��������ҲΪ7.3g���������������Ƶ�����ΪY����ϻ�ѧ����ʽ2HCl+Ca(OH)2==CaCl2+2H2O�����Ȼ�����������ɼ�����������Ƶ�������
��100%=80%����4���ɣ�2����ѧ����ʽ����ɵ÷�Ӧ���Ȼ�������Ϊ7.3g����Ϊ��Ӧ�ٽ�������������պ÷�Ӧ��һ�룬����Һa���Ȼ��������ҲΪ7.3g���������������Ƶ�����ΪY����ϻ�ѧ����ʽ2HCl+Ca(OH)2==CaCl2+2H2O�����Ȼ�����������ɼ�����������Ƶ�������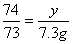 ��y=7.4g����5����Һb������Ϊ�Ȼ��ƣ���Ӧ�ٺͷ�Ӧ�ھ������Ȼ��ƣ����ã�2���л�ѧ����ʽ���Լ������Ӧ�����ɵ��Ȼ�������Ϊ11.1g�����ã�4���еĻ�ѧ����ʽ���Ȼ�����������Լ�����Ȼ��Ƶ�����z��
��y=7.4g����5����Һb������Ϊ�Ȼ��ƣ���Ӧ�ٺͷ�Ӧ�ھ������Ȼ��ƣ����ã�2���л�ѧ����ʽ���Լ������Ӧ�����ɵ��Ȼ�������Ϊ11.1g�����ã�4���еĻ�ѧ����ʽ���Ȼ�����������Լ�����Ȼ��Ƶ�����z��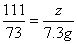 ��z=11.1g���������غ㶨�ɿɵã�������Һ����Ϊ80g+10g-4.4g+107g+7.4g=200g��������Һ����������Ϊ
��z=11.1g���������غ㶨�ɿɵã�������Һ����Ϊ80g+10g-4.4g+107g+7.4g=200g��������Һ����������Ϊ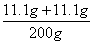 ��100%=11.1%����6����Ӧ������պ÷�Ӧ��һ�룬��80g�����к����Ȼ�������Ϊ7.3g��2=14.6g�������ˮ������Ϊm��������Һϡ��ǰ��������������ɵã���80g-m����36.5%=14.6g������ó�m=40g��
��100%=11.1%����6����Ӧ������պ÷�Ӧ��һ�룬��80g�����к����Ȼ�������Ϊ7.3g��2=14.6g�������ˮ������Ϊm��������Һϡ��ǰ��������������ɵã���80g-m����36.5%=14.6g������ó�m=40g��
���㣺 ��ѧ����ʽ���㣻�����غ㶨�ɣ�ϡ����Һ

ʹ���ܶ�С��ǿ�ȴ��þ�Ͻ��ܼ����������أ��Ӷ������������ĺͷ����ŷš�
|
��2����ҵ��þ��һ��ԭ����2MgO+Si+2CaO ====== 2Mg��+Ca2SiO4�����������еĿ����Է�Ӧ�дٽ����ã����ܷ�ֹ�����е�������Mg��Ӧʹ��Ʒ���� ���ѧʽ����
��3����þ��ԭ��MgO�ɴӺ�ˮ�л�á�С������þ�����ᷴӦ��ķ�Һ��ģ��Ӻ�ˮ�л�ȡMgO�Ĺ��̣�ʵ�����£�
����1����������Һ�У��߽���߷�������CaO����MgCl2��ȫ����Ϊֹ�����˵�Mg(OH)2���塣������8.4g CaO��
����2����Mg(OH)2������ȷֽ�ΪMgO��ˮ������MgO������Ϊ4.0g ��
�ٲ���2��Mg(OH)2�ֽ�Ļ�ѧ����ʽΪ ��
��ͨ������MgO����������������Һ�к�MgCl2������m= g��
�۷���ʵ�����ݣ���֪������з����Ļ�ѧ��Ӧ�У�
CaO+H2O==Ca(OH)2��Ca(OH)2+MgCl2==CaCl2+Mg(OH)2���� ��
 D����ʾһ����������������һ����ԭ�Ӻ�������ԭ�ӹ���
D����ʾһ����������������һ����ԭ�Ӻ�������ԭ�ӹ��� ��ͼΪij��Ӧ��ͬһ�����з�Ӧǰ����ʾ��ͼ������˵������ȷ����( )
��ͼΪij��Ӧ��ͬһ�����з�Ӧǰ����ʾ��ͼ������˵������ȷ����( )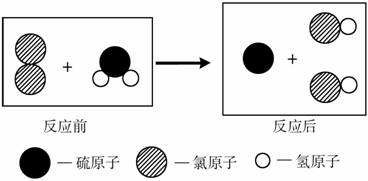

 ��
��