��Ŀ����
32����Ӫ����ɫ���У��ǻ������������������ά�ֺ���Ŀɳ�����չ��ÿ�����������ʥְ����ش�������ں���ļ������⣺
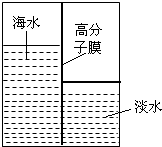
��1����ˮ����Դ�ḻ���������Ľ�����������
��2���γ���ˮɹ�ε�ԭ����
A���Ȼ��Ƶ��ܽ�����¶�Ӱ��仯�ϴʲ�ȡ���½ᾧ�ķ���
B�����Ȼ����ܽ�����¶�Ӱ��仯���ʲ�ȡ����ˮ�ֵķ���
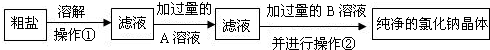
��3�����ô�������Ҫ����ɰ���Ȼ�þ�����ʣ��ֶ����ᴿ����Ҫ�����������£�

�ٲ���I��������
��A��Һ��
A��NaOH B��KOH C��H2SO4 D��HCl��
��1����ˮ����Դ�ḻ���������Ľ�����������
Na+
������ţ����������ķ�����H2O
������ţ�����2���γ���ˮɹ�ε�ԭ����
B
��A���Ȼ��Ƶ��ܽ�����¶�Ӱ��仯�ϴʲ�ȡ���½ᾧ�ķ���
B�����Ȼ����ܽ�����¶�Ӱ��仯���ʲ�ȡ����ˮ�ֵķ���
��3�����ô�������Ҫ����ɰ���Ȼ�þ�����ʣ��ֶ����ᴿ����Ҫ�����������£�

�ٲ���I��������
����
����A��Һ��
A
��B��Һ��D
����ѡ����ʵ������գ�A��NaOH B��KOH C��H2SO4 D��HCl��
��������1�����ݺ�ˮ�е���Ҫ���ʷ�����
��2�������Ȼ��Ƶ��ܽ�����¶�Ӱ��������ٽ��ʵ���ҳ�������������
��3�������ᴿ�������ܽ��Ҫ�ù��˵ķ�����ȥ��ɳ��Ȼ��������������������Һ��ȥ�����Ȼ�þ��Ȼ��������ȥ����������������Һ��
��2�������Ȼ��Ƶ��ܽ�����¶�Ӱ��������ٽ��ʵ���ҳ�������������
��3�������ᴿ�������ܽ��Ҫ�ù��˵ķ�����ȥ��ɳ��Ȼ��������������������Һ��ȥ�����Ȼ�þ��Ȼ��������ȥ����������������Һ��
����⣺��1����Ϊ��ˮ��������ѧ����������H2O����ξ���NaCl��
�ʴ�Ϊ��Na+��H2O��
��2����Ϊ�Ȼ��Ƶ��ܽ�����¶�Ӱ��Ƚ�С������Ӧ�������ᾧ�ķ���ʹ��������������Ӧѡ��B��
�ʴ�Ϊ��B��
��3���Ѵ����ܽ��Ҫ�ù��˵ķ���ȥ��������ˮ����ɳ��Ȼ���Ҫ���������A����������Һ��ȥ�������Ȼ�þ�����˺��ټ���Bϡ�����ȥ����������������Һ��
�ʴ�Ϊ���ٹ��ˣ���AD��
�ʴ�Ϊ��Na+��H2O��
��2����Ϊ�Ȼ��Ƶ��ܽ�����¶�Ӱ��Ƚ�С������Ӧ�������ᾧ�ķ���ʹ��������������Ӧѡ��B��
�ʴ�Ϊ��B��
��3���Ѵ����ܽ��Ҫ�ù��˵ķ���ȥ��������ˮ����ɳ��Ȼ���Ҫ���������A����������Һ��ȥ�������Ȼ�þ�����˺��ټ���Bϡ�����ȥ����������������Һ��
�ʴ�Ϊ���ٹ��ˣ���AD��
�����������ʵ��ܽ�����¶�Ӱ�첻��ʱ�ü��������ķ���ʹ��ᾧ�����������ʵ��ܽ�����¶�Ӱ��Ƚϴ�ʱ����ȴ�ȱ�����Һ�ķ���ʹ��ᾧ������

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ
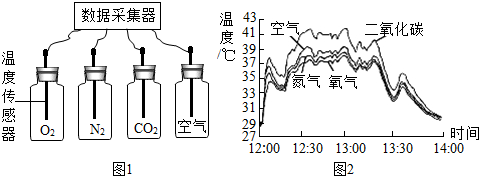
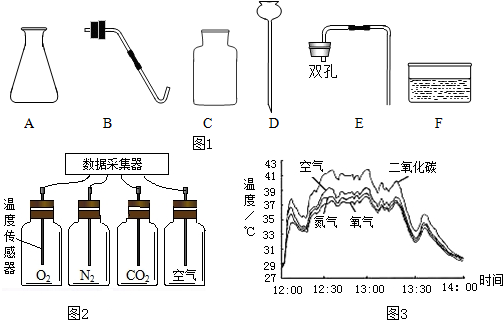
 ������Ӧ�����е�
������Ӧ�����е� �������绷���յ������ǡ�Ӫ����ɫ���У��ǻ����������ij��ѧ̽��С��Ϊ̽��Ӱ�����������ЧӦ�������أ�����̫����������N2��O2��CO2����������ЧӦǿ�ȵıȽ�ʵ�飮����������ǵ�̽�����̣���д�йؿո�
�������绷���յ������ǡ�Ӫ����ɫ���У��ǻ����������ij��ѧ̽��С��Ϊ̽��Ӱ�����������ЧӦ�������أ�����̫����������N2��O2��CO2����������ЧӦǿ�ȵıȽ�ʵ�飮����������ǵ�̽�����̣���д�йؿո�