��Ŀ����
��ͼ��ʾijЩ���ʼ�ת����ϵ��
A��E��GΪ���壬��A��G��Ϊ���ʣ�
EΪ����ɫ����,GΪ����������C��F��
D��HΪ��ɫ���壬����C��F������
ͬԪ����ɵ���ɫ���壬��C�ж���
BΪ��������ɫҺ�壬IΪ�л����
�ش��������⣺
��1��E�Ļ�ѧʽ�� ��
��2������Ȼ�����ձ���ڷ�Ӧ�ݣ�������Ϊ ��
��3���ڹ�ҵ������Ϊ��ֹ��Ӧ�ܵķ������ɲ�ȡ�Ĵ�ʩ�� ��дһ�㼴�ɣ���
��4��ͨ����Ӧ�ۿ���˵��B���� ������ĸ����
A��������Ԫ�غ���Ԫ����� B������������������ӹ���
��5����Ӧ�ٵĻ�ѧ����ʽ�� ��

(1)Fe2O3 (2)������� (3) ˢ���� (4)A (5) C+H2O==CO +H2

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
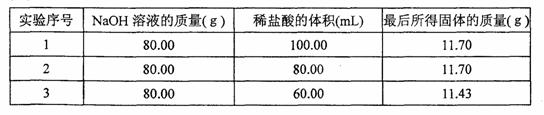
Сѧ��10����Ӧ����ϵ�д���ͼ��θ������θҩ�IJ��ֱ�־��θҩ���������������к�θ������θ��(��Ҫ������)��ij���߰���־�ϵķ��÷�����ҩ��������ת�����㻼���������������õĴ�θҩ��(������ȡ����)��
| θ������ ���ܣ�����θ����������θ�� �ɷ֣�ÿƬ��������þ250 mg ���÷�����ÿ�����Σ�ÿ��2Ƭ |
(1)����������þ������Ϊ____________ mg��
(2)�����Ͽ��к�θ���е�HCl������Ϊ____________ mg��
 ��2�������к�����ɳ���������ʺ��Ȼ��Ƶȿ��������ʣ�ijѧϰС����Ʒ����Ȼ��ƺ��Ȼ��ƹ��巽�����£�
��2�������к�����ɳ���������ʺ��Ȼ��Ƶȿ��������ʣ�ijѧϰС����Ʒ����Ȼ��ƺ��Ȼ��ƹ��巽�����£� .̼泥�NH4 HCO3��
.̼泥�NH4 HCO3�� ��Ҫ֤������ˮ��Ӳˮ������ˮ������ �����顣
��Ҫ֤������ˮ��Ӳˮ������ˮ������ �����顣