��Ŀ����
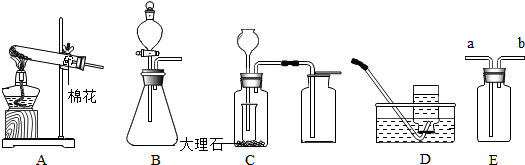
10����ѧ��ȤС���ijƷ��������̼��ƺ�����������̽����������Ħ������Ҫ��̼��ơ�����������ɣ������ɷ���������ʱ���������ɣ���������ͼ��ʾװ�ã�ͼ�мг�������ȥ������ʵ�飬��ַ�Ӧ�ⶨC�����ɵ�BaCO3������������ȷ��̼��Ƶ�������������ش��������⣮����ʵ����̻ش��������⣺
��1��ʵ��������Ⱥ����ι���������ڶ��ι��������Ŀ�������ϲ�����װ���е�CO2��ʹ���ɵ�CO2��ȫ�����գ�
��2��д��C�еĻ�ѧ����ʽ��CO2+Ba��OH��2�TBaCO3��+H2O��
��3��ʵ����ȷ��ȡ������Ʒ��10.00g���������βⶨ�����BaCO3ƽ������Ϊ3.94g������Ʒ��̼��Ƶ���������Ϊ20%��
��4��ijͬѧ�����C��Ba��OH��2��Һ����NaOH��Һ��ͨ���ⶨCװ�÷�Ӧǰ���������Ҳ���ԲⶨCaCO3�ĺ�����ʵ��֤�����˲ⶨ�Ľ��ƫ�ߣ�ԭ����B�е�ˮ�������Ȼ�������Ƚ���װ��C�У�
���� ��1��ʵ��ͨ��Cװ�����ɵ�̼�ᱵ�������ⶨ������̼����������������������̼��Ƶ�����������װ���в������ֶ�����̼�����ܱ���ȫ���գ����²ⶨ��̼�ᱵ������ƫС��
��2��������̼������������Ӧ����̼�ᱵ��ˮ��
��3������̼��������ᷴӦ���ɶ�����̼��������̼��������������̼�ᱵ�����ɽ���̼�����̼�ᱵ�Ĺ�ϵʽ����������е�������ϵ������ɣ�
��4��B�е�ˮ�������Ȼ�������Ƚ���װ��C��
��� �⣺��1��װ���в������ֶ�����̼�����ܱ���ȫ���գ����²ⶨ��̼�ᱵ������ƫС����������ͨ�����������Ϊ�������ɵ�CO2����ȫ������C�У�ʹ֮��ȫ��Ba��OH��2��Һ���գ����ϲ�����װ���е�CO2 ��ʹ���ɵ�CO2��ȫ�����գ�
�ʴ�Ϊ�����ϲ�����װ���е�CO2 ��ʹ���ɵ�CO2��ȫ�����գ�
��2��������̼������������Ӧ����̼�ᱵ��ˮ����Ӧ����ʽΪCO2+Ba��OH��2�TBaCO3��+H2O���ʴ�Ϊ��CO2+Ba��OH��2�TBaCO3��+H2O��
��3������̼��������ᷴӦ���ɶ�����̼��������̼��������������̼�ᱵ�����ķ���ʽ�ֱ��ǣ�CaCO3+2HCl�TCaCl2+H2O+CO2����Ba��OH��2+CO2�TBaCO3��+H2O�����Կɽ���CaCO3��CO2��BaCO3�Ĺ�ϵʽ������̼��Ƶ���������Ϊx��
CaCO3��CO2��BaCO3
100 197
10g��x 3.94g
��$\frac{100}{10g��x}=\frac{197}{3.94g}$�����x=20%��
�ʴ�Ϊ��20%��
��5��B�е�ˮ�������Ȼ�������Ƚ���װ��C�У����²ⶨ������̼������ƫ�ⶨ��̼��Ƶ�����ƫ��̼��Ƶ���������ƫ�ߣ�
�ʴ�Ϊ��B�е�ˮ�������Ȼ�������Ƚ���װ��C�У�
���� ���⿼���ʵ��ԭ���������������⼰���۵ȣ��ѶȽϴ��Ƕ�����֪ʶ���ۺ����ã���Ҫѧ��������ʵ�Ļ���֪ʶ��������⡢������������������ʵ��ԭ���ǽ��Ĺؼ���

 ���ٴ�����ɽ����ϵ�д�
���ٴ�����ɽ����ϵ�д� ���ٴ���������ѧϰ����ѧ�ں����ν�ϵ�д�
���ٴ���������ѧϰ����ѧ�ں����ν�ϵ�д�| ѡ�� | ���ʣ�������Ϊ���ʣ� | �����Լ����������� |
| A | FeSO4��CuSO4�� | ����������ˮ���������������ۣ����ˣ������ᾧ |
| B | NaOH��Һ��Na2CO3�� | ������������������Һ������ |
| C | ͭ�ۣ���������ĩ�� | ��������ϡ���ᣬ���ˣ�ϴ�ӣ����� |
| D | N2��O2�� | ������ͨ�����ȵ�̿�� |
| A�� | A | B�� | B | C�� | C | D�� | D |
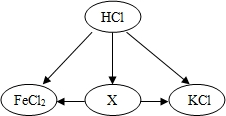 ��ͼΪ�������ʵ��ת����ϵ������ͼʾ��ϵ�жϣ�X�Ļ�ѧʽ�����ǣ�������
��ͼΪ�������ʵ��ת����ϵ������ͼʾ��ϵ�жϣ�X�Ļ�ѧʽ�����ǣ�������| A�� | Na2CO3 | B�� | CuCl2 | C�� | Ca��NO3��2 | D�� | NaCl |
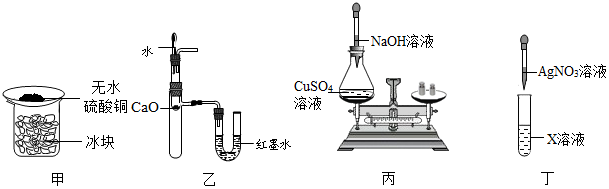
| A�� | ��װ����֤��������ˮ�� | B�� | ��װ����֤CaO��ˮ��Ӧ����ЧӦ | ||
| C�� | ��װ����֤�����غ㶨�� | D�� | ��װ����֤X��Һ���Ƿ���Cl- |
| A�� | ������Һ��ʯ������ȼ�� | B�� | �ƹ�ʹ��̫������ˮ�� | ||
| C�� | ������ú�������� | D�� | ��ֹ��˽�ҳ����� |
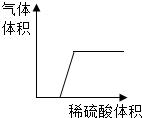 ��HCl��NaOH��Na2CO3��Ca��OH��2��Һ��ȡ�����е����ֻ�ϣ�
��HCl��NaOH��Na2CO3��Ca��OH��2��Һ��ȡ�����е����ֻ�ϣ� ���㽭���ߡ�ˮ����𣬿��������ⳡ�꣬ȴ��һ����ֵ��г䵱�ˡ��͡��Ľ�ɫ��������30�ҳ�һ·�������֡����ϴ�������ʯ��13Сʱ!��һ�ж�����Ϊһ������20��ֵ�ʯ�Ĵ�������ݽ��ܣ���ʯ����ѧ����̼���ƣ�CaC2��������ȼ����һ���л��ϳɻ�ѧ��ҵ�Ļ���ԭ�ϣ���ѧ���ʷdz����ã���ˮ���ܼ��ҷֽ������Ȳ��C2H2��������������ƣ��ͷų��������ȣ���Ȳ���岻������ˮ���ܶȱȿ���С����ش�
���㽭���ߡ�ˮ����𣬿��������ⳡ�꣬ȴ��һ����ֵ��г䵱�ˡ��͡��Ľ�ɫ��������30�ҳ�һ·�������֡����ϴ�������ʯ��13Сʱ!��һ�ж�����Ϊһ������20��ֵ�ʯ�Ĵ�������ݽ��ܣ���ʯ����ѧ����̼���ƣ�CaC2��������ȼ����һ���л��ϳɻ�ѧ��ҵ�Ļ���ԭ�ϣ���ѧ���ʷdz����ã���ˮ���ܼ��ҷֽ������Ȳ��C2H2��������������ƣ��ͷų��������ȣ���Ȳ���岻������ˮ���ܶȱȿ���С����ش�