��Ŀ����
������ĩʱ�䣬ij����֯Ұ�����
��1������ʱ�Ƚ�ֽ��ȼ������ȼ�ŵ�ֽ��ȼ��֦��˵��ֽ�ŵ��Ż��Ͽ�֦���Ż�� ����ߡ��͡���
��2������֦������ʱ��������Ũ�̣�����Ϩ�𣬳�����ֿ�֦�����յø�����˵��ȼ�ϳ��ȼ����Ҫ�������� ��
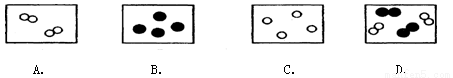
��3��ijͬѧ����һ��ֽ���������ͼ��ʾ��������ֽ�������ֽ�������������������ʢ�����ϣ���ȼ��ȼ��ʱֽ���ᱻ��ȼ��ֽ���ʹ��ʱֽ���ᱻ��ȼ��ԭ���� ��

A���������㣬ֽ��ȼ��
B��ֽ�ű�ˮ��ʪ���Ż�㽵����
C��ֽ�������ʱ���Ż��������
D��ֽ����е�ˮ����ʱ���ȣ��¶ȴﲻ��ֽ���Ż��
ֽ�����ȼ���ǹ���ƾ�������ƾ�����Ҫ�ɷ����Ҵ���C2H5OH����д���ƾ����ȼ�յĻ�ѧ����ʽ�� ��
��4��Ұ��������ͬѧ�ѻ�Ϩ��������ҽ��ϵ�ˮ�������ԭ����������Ŀ���� ��
��1���� ��2����ȼ����������ֽӴ� ��3��D C2H5OH+3O2��ȼ2CO2+3H2O
��4�� ���¶Ƚ��͵���ȼ���Ż�����£���ֹ��ȼ
��������
�����������1������ʱ�Ƚ�ֽ��ȼ������ȼ�ŵ�ֽ��ȼ��֦��˵��ֽ�ŵ��Ż��Ͽ�֦���Ż���
��2������֦������ʱ��������Ũ�̣�����Ϩ�𣬳�����ֿ�֦�����յø����������������������ĽӴ������˵��ȼ�ϳ��ȼ����Ҫ�������ǿ�ȼ����������ֽӴ�
��3��ȼ�յ���������1����ȼ�2�����������������3���¶ȴﵽ�Ż�㣻ֽ���ʹ��ʱֽ���ᱻ��ȼ��ԭ����ֽ����е�ˮ����ʱ���ȣ��¶ȴﲻ��ֽ���Ż�㣬ѡD ���ƾ����ȼ�յĻ�ѧ����ʽ�� C2H5OH+3O2��ȼ2CO2+3H2O
��4������ԭ������1��������ȼ�2���������������������3�����µ��Ż�����£�Ұ��������ͬѧ�ѻ�Ϩ��������ҽ��ϵ�ˮ�������ԭ����������Ŀ���ǣ����¶Ƚ��͵���ȼ���Ż�����£���ֹ��ȼ
���㣺ȼ�յ�����������ԭ����ȼ�յ�����