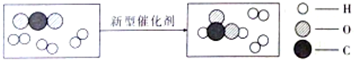
��Ŀ����
3��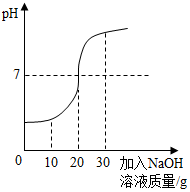 ��ѧ��ȤС���ij��ҵ��ˮ������ΪHCl��NaCl���е�HCl�������вⶨ���ס�����λͬѧ���ṩ��ͬ�IJⶨ������
��ѧ��ȤС���ij��ҵ��ˮ������ΪHCl��NaCl���е�HCl�������вⶨ���ס�����λͬѧ���ṩ��ͬ�IJⶨ��������1����ͬѧ������кͷ�
ȡ100g��ˮ���ձ��У���ε���������������Ϊ20%��NaOH��Һ����Ӧ��������Һ��pH�仯��ͼ��ʾ�����ˮ��HCl������������д����ϸ�ļ�����̣���
��2����ͬѧ��������
����AgNO3��Һ����NaOH��Һ���������ɳ�����������ȷ����ˮ��HCl����������������Ϊ�����ƫ���ƫ��ƫС������Ӱ�족����
���� �Ȼ��Ʋ��ܺ��������Ʒ�Ӧ��ϡ�����ܺ��������Ʒ�Ӧ�����Ȼ��ƺ�ˮ��
�������ܺ������ӽ�ϳɰ�ɫ�����Ȼ�����
��� �⣺��1����ͼ��֪��������20g 20%��NaOH��Һʱ����Һ��pH=7��˵����������ǡ����ȫ��Ӧ����100g��Һ�к���HCl������Ϊx��
HCl+NaOH�TNaCl+H2O��
36.5 40
x 20g��20%
$\frac{36.5}{x}$=$\frac{40}{20g��20%}$��
x=3.65g��
��ˮ��HCl����������Ϊ��$\frac{3.65g}{100g}$��100%=3.65%��
�𣺷�ˮ��HCl����������Ϊ3.65%��
��2������AgNO3��Һ����NaOH��Һ���������ɳ�����������ȷ����ˮ��HCl�����������������ƫ��������Ϊ��Һ�е��Ȼ��⡢�Ȼ��ƶ��ܹ�����������Ӧ�����Ȼ������������ɳ�����������������Ȼ�������ƫ�Ӷ����·�ˮ��HCl����������ƫ��
���ƫ��
���� ������Ҫ����ѧ�����ü��跨�ͻ�ѧ����ʽ���м�����ƶϵ�����������ʱҪע��淶�Ժ�ȷ�ԣ�

��ϰ��ϵ�д�
�����Ŀ
14��С���������ɵó������й��ɺ������ǣ�������
| A�� | pH����7����Һһ���Ǽ���Һ | B�� | ���������һ�������� | ||
| C�� | ����̼Ԫ�صĻ�����һ�����л��� | D�� | �ᡢ����һ��������Ԫ�� |
18�������������֮�����ϵ�ǻ�ѧѧ�����е�˼ά��ʽ�����жԺ����ʵ���۽��ʹ�����ǣ�������
| A�� | 50mL�ƾ���50mLˮ��Ϻ����С��100mL������Ϊ���ӱ�С�� | |
| B�� | ��ԮȮ�ܸ�����ζ�������������ڵ��µ�������������Ϊ�����ڲ����˶� | |
| C�� | ���ˮ��������������������Ϊ�����ڻ�ѧ�仯�п����ٷ� | |
| D�� | ��ѧ��Ӧ���������غ㶨�ɣ�����Ϊ��Ӧǰ��ԭ�Ӹ�����ԭ������� |
8�����ɷ���ѧϰ��ѧ����Ч�ķ���֮һ�����жԲ��ֻ�ѧ֪ʶ�Ĺ�����ȷ��һ���� ��������
| A����������ʶ | B���Գ�ʶ����ʶ |
| ���Ȼ����������Ӻ������ӹ��� �ھ�����ͬ����������������ͬ��Ԫ�� �۴�������Ӳ�һ�������� | ��ϴ�Ӽ���ȥ�������黯���� ������п�����������е���Ԫ�� ����ˮ����ԭ���ǽ��Ϳ�ȼ����Ż�� |
| C���Լ������ʶ | D���Ը�������� |
| ������Ӳˮ����ˮ���ӷ���ˮ �����ֹ���������Һ��ˮ���Ӷ������� ����������������Һ�ͳ����ʯ��ˮ����CO2 | ��ʹ��ɫʯ����Һ�����IJ�һ���Ǽ���Һ ���Ȼ�����ľ�һ��ʹ�ã��������ӷ�Ч �����ɵ��ʺͻ�����ķ�Ӧһ�����û���Ӧ |
| A�� | A | B�� | B | C�� | C | D�� | D |
12�����и��������У��ܴ���������ǣ�������
| A�� | H+��SO42-��OH-��Ba2+ | B�� | Na+��K+��Cl-��SO42- | ||
| C�� | Na+��H+��NO3-��CO32- | D�� | K+��Mg2+��NO3-��OH- |
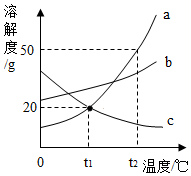 ��ͼ��a��b��c�������ʵ��ܽ�����ߣ����ͼ�ش��������⣺
��ͼ��a��b��c�������ʵ��ܽ�����ߣ����ͼ�ش��������⣺