��Ŀ����
5��Ҫʵ�֡���ѧ����������á������뱣��������������̼����Իش��������⣺��1��������һ������Ⱦ�����ϣ��о����֣�������-��ѭ��ˮ��������������Ҫ�漰�����������ԣ���
��SO2+2H2O+I2=H2SO4+2HI��
��2HI=H2��+I2��
��2H2SO4=2SO2��+O2��+2H20
��ش�ѭ���ֽ�ˮ�Ĺ����в���O2��H2��������Ϊ8��1ʵ������п��ϡ���ᷴӦ�Ļ�ѧ����ʽ��Zn+H2SO4�TZnSO4+H2��
��2��ȼ��ȼ��ʱ������������ͬ����֪��ij�¶��¸���ȼ��ȼ��ʱ�������������±���
| ȼ�� | ����/g | ���� | ����������/KJ |
| ���� | 2 | ˮ���� | 241.8 |
| ���� | 16 | ������̼�����ˮ���� | 890.3 |
| �Ҵ� | 46 | ������̼�����ˮ���� | 1367 |
���� ��1������ʵ������п��ϡ������ȡ�����ķ�Ӧд����Ӧ�Ļ�ѧ����ʽ������ѭ���ֽ�ˮ�ķ�Ӧ�������O2��H2�������ȣ�
��2�����ݱ������ݼ���Ƚϣ�
��� �⣺��1��ʵ������п��ϡ������ȡ��������Ӧ�Ļ�ѧ����ʽ�ǣ�Zn+H2SO4�TZnSO4+H2������ѭ���ֽ�ˮ�ķ�Ӧ��֪�����ɵ�O2��H2�Ĺ�ϵ��O2��2H2�������ɵ�O2��H2����������8��1��
��2���ɱ��������ݿ�֪��ÿ1gȼ��ȼ��ʱ�����������ֱ��ǣ�
H2��$\frac{241.8KJ}{2}$=140.9KJ��CH4��$\frac{890.3KJ}{16}$=55.6KJ��CH3CH2OH��$\frac{1367K}{46}$=29.7KJ
�����ϼ����֪��ȼ�յ�λ����������ȼ�ϲ����������ɶൽ�ٵ�˳����H2��CH4��CH3CH2OH��
�ʴ�Ϊ��8��1��Zn+H2SO4�TZnSO4+H2����H2��CH4��CH3CH2OH
���� ��Դ����Ϣ�����ϡ������ǵ��������Ĵ���Ҫ���⣬����ԴΣ���������صĽ��죬����������ȵ㣬Ҳ�ǻ�ѧ������ȵ㣬��Ҫ������Դ�ķ��ࡢ��Դʹ�öԻ�����Ӱ�졢����Դ�����ļ�����ǰ���ȣ�

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ
20����ͼ��һ��ʱ���ɹ۲쵽��������

| A�� | B����Һ��� | B�� | A����Һ��� | C�� | A��B������ | D�� | A��B����� |
4�������ĸ�ͼ�ֱ��Ӧ���ֲ������̣�������ȷ���ǣ�������
| A�� | 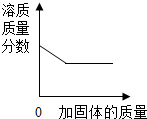 ij�¶��£���һ�������ı����Ȼ�����Һ�м����Ȼ��ƹ��� | |
| B�� | 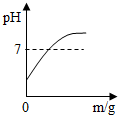 Ũ���᳨�ڷ��� | |
| C�� |  ��������һ�������NaOH��Һ�м���ϡ���� | |
| D�� |  ��HCl��CuCl2���Һ�еμ�KOH��Һ |
5��ʵ��֤��M�����ڿ�����ȼ������CO2���ɣ������ݴ˽�������ж�M�ijɷ��Ʋ���ȷ���ǣ�������
| A�� | M��һ����̼ | B�� | M�Ǽ��� | ||
| C�� | M��һ������̼Ԫ�� | D�� | M�к���̼����Ԫ�� |

 ��ͼ��ʾ����һƿ������ˮע��Һ��ǩ������֪�����ܶȽ���Ϊ1g/cm3�����ô�ƿ��������Һ������벡�����ڵ��Ȼ���Ϊ���ٿˣ�
��ͼ��ʾ����һƿ������ˮע��Һ��ǩ������֪�����ܶȽ���Ϊ1g/cm3�����ô�ƿ��������Һ������벡�����ڵ��Ȼ���Ϊ���ٿˣ�