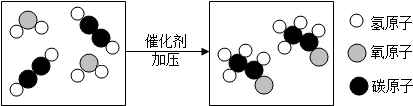
��Ŀ����
19�� ij������Ʒ�к�������NaCl��С��ͬѧȡ����Ʒ23gȫ���ܽ���150gˮ�У�����μ���160gϡ���ᣬ��Ӧ�в���������������������������ϵ��ͼ��ʾ������ʾ����ط�ӦΪNa2CO3+2HCl�T2NaCl+H2O+CO2����
ij������Ʒ�к�������NaCl��С��ͬѧȡ����Ʒ23gȫ���ܽ���150gˮ�У�����μ���160gϡ���ᣬ��Ӧ�в���������������������������ϵ��ͼ��ʾ������ʾ����ط�ӦΪNa2CO3+2HCl�T2NaCl+H2O+CO2�������㵱ǡ����ȫ��Ӧʱ��
��1�������Ķ�����̼��������8.8�ˣ�
��2����ʱ���õ�����Һ�����ʵ�����������д����Ҫ�ļ�����̣����������0.1%��
���� �������е�֪ʶ���з�����𣬾�ͼ����֪�����ɵĶ�����̼�����������ݶ�����̼����������μӷ�Ӧ��̼���Ƶ����������ɵ��Ȼ��Ƶ�������Ȼ�������������������ʽ���㼴�ɣ�
��� �⣺��1����ͼ���Կ��������ɵĶ�����̼������Ϊ8.8g�����8.8��
��2����̼���Ƶ�����Ϊx�����ɵ��Ȼ��Ƶ�����Ϊy
Na2CO3+2HCl�T2NaCl+H2O+CO2��
106 117 44
x y 8.8g
$\frac{106}{x}=\frac{117}{y}=\frac{44}{8.8g}$
x=21.2g y=23.4g
��Ʒ���Ȼ��Ƶ�����Ϊ��23g-21.2g=1.8g
�ʷ�Ӧ���Ȼ�����Һ��������������Ϊ��
$\frac{1.8g+23.4g}{23g+150g+135.8g-8.8g}��100%=8.4%$
�����õ�����Һ�����ʵ���������Ϊ8.4%��
���� ������Ҫ���黯ѧ����ʽ����д���йػ�ѧ����ʽ�ļ��㣬��ɴ��⣬�����������е�֪ʶ���У�

��ϰ��ϵ�д�
�����Ŀ
10��Ŀǰ�г��ϳ��۵ġ��ӵ�ʳ�Ρ������߸�ţ�̡������������͡��ȣ���Щ�⡢�ơ�����ָ��������
| A�� | ԭ�� | B�� | ���� | C�� | Ԫ�� | D�� | ���� |
7�����������е��������ʣ�������Ϊ���ʣ���ֻ������NaOH�Ĺ������Һ���ܳ�ȥ���ǣ�������
| A�� | CO2��H2O�� | B�� | CO2��HCl�� | C�� | NaCl��Һ��MgCl2�� | D�� | KNO3��Һ��H2SO4�� |
4�����һ���Ż���ʵ�鷽������֤ij�Ȼ�����Һ�л���̼���ơ������ƺ��������Ʋ��ᴿ�Ȼ��ƣ���ʵ�����̼�����ʵ��������ͼ��ʾ������˵������ȷ���ǣ�������
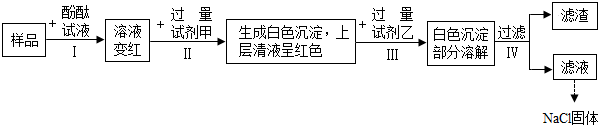

| A�� | ��Ʒ��Һ��pH����7 | |
| B�� | �Լ�����BaCl2��Һ | |
| C�� | �ڢ�ʵ���л��۲쵽�������������ݲ��� | |
| D�� | ���һ��������Һ���е�������NaCl��BaCl2��HCl��ָʾ�����⣩ |
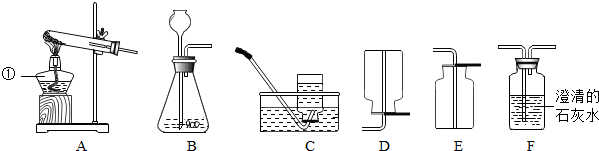
11������ʵ���������̲��������ǣ�������
| A�� | 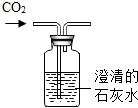 ����CO2���� | B�� | 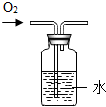 ������������ | ||
| C�� |  ��ȡ10.5g���Ȼ��ƹ��� | D�� |  ���� |
8��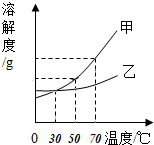 �ס����������ʵ��ܽ��������ͼ��ʾ��70��ʱ�����������ļס������ֹ��壨�����ᾧˮ���ֱ���뵽��ʢ100gˮ�������ձ��У���ֽ���������ȫ�ܽ⣬���µ�50�棬�۲쵽����Һ�й�������������˵������ȷ���ǣ�������
�ס����������ʵ��ܽ��������ͼ��ʾ��70��ʱ�����������ļס������ֹ��壨�����ᾧˮ���ֱ���뵽��ʢ100gˮ�������ձ��У���ֽ���������ȫ�ܽ⣬���µ�50�棬�۲쵽����Һ�й�������������˵������ȷ���ǣ�������
 �ס����������ʵ��ܽ��������ͼ��ʾ��70��ʱ�����������ļס������ֹ��壨�����ᾧˮ���ֱ���뵽��ʢ100gˮ�������ձ��У���ֽ���������ȫ�ܽ⣬���µ�50�棬�۲쵽����Һ�й�������������˵������ȷ���ǣ�������
�ס����������ʵ��ܽ��������ͼ��ʾ��70��ʱ�����������ļס������ֹ��壨�����ᾧˮ���ֱ���뵽��ʢ100gˮ�������ձ��У���ֽ���������ȫ�ܽ⣬���µ�50�棬�۲쵽����Һ�й�������������˵������ȷ���ǣ�������| A�� | ���µ�50��ʱ�ҵ���Һһ���й������� | |
| B�� | 70��ʱ���ü��ҵ���Һһ�����Dz�������Һ | |
| C�� | ����50���ٽ��µ�30�棬���ҵ���Һ�������������� | |
| D�� | ���µ�30�������Һ�����ʵ������������ |
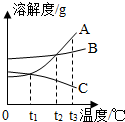 ��ͼ��A��B��C�������ʵ��ܽ�����ߣ���ͼ�ش�
��ͼ��A��B��C�������ʵ��ܽ�����ߣ���ͼ�ش�