题目内容
7.某同学去某风景区游玩时,取回了若干块矿石样品,他采用了以下的方法对样品中碳酸钙的质量分数进行检测:取这种石灰石样品6g,把40g稀盐酸四次加入,测量过程所得数据见下表(已知石灰石样品中含有的杂质不溶于水,不与盐酸反应).| 加入稀盐酸的次序 | 1 | 2 | 3 | 4 |
| 加入稀盐酸的质量/g | 10 | 10 | 10 | 10 |
| 剩余固体的质量/g | 4.0 | m | 0.6 | 0.6 |
(2)样品中碳酸钙的质量分数是90.0%.
(3)所用稀盐酸的质量分数.(计算过程)
分析 (1)比较第一次和第三次的数据可知第一次中盐酸完全反应,消耗碳酸钙6.0g-4.0=2.0g,因此第二次中也是消耗2.0g碳酸钙,故可以求出m的值;
(2)由杂质的质量可以求出碳酸钙的质量,进而求出碳酸钙的质量分数;
(3)根据题意可以知道第一次盐酸和2g碳酸钙恰好完全反应,可以据此求出盐酸的质量分数.
解答 解:(1)比较第一次和第三次的数据可知,第一次加入10g盐酸后剩余固体的质量为4.0g,而第三次加入稀盐酸后剩余固体的质量为0.6g,所以可以判断第一次中盐酸完全反应,消耗碳酸钙6.0g-4.0=2.0g,即10g 稀盐酸能够消耗2g碳酸钙,因此第二次中也是消耗2.0g碳酸钙,故可以求出m=4.0-2.0=2.0;
(2)碳酸钙的质量为:6.0g-0.6g=5.4g,故其质量分数为:$\frac{5.4g}{6.0g}$×100%=90.0%
(3)设盐酸的质量为X,则:
CaCO3+2HCl═CaCl2+H2O+CO2↑
100 73
2g X
$\frac{100}{2g}=\frac{73}{X}$
X=1.46 g.
所以盐酸的质量分数为:$\frac{1.46g}{10g}$×100%=14.6%.
故答案为:(1)2.0;
(3)90.0%;
(4)14.6%.
点评 本题主要考查学生运用化学方程式和质量分数公式进行计算的能力,解答本题时学生需要认真分析图表数据,根据物质反应时的质量关系,正确运用公式和化学方程式进行解答.

练习册系列答案
相关题目
17.现有Fe2O3、ZnO、CuO的混合固体ag,在高温条件下与足量CO充分反应后,得到金属混合物4.6g.将生成CO2用足量的澄清石灰水吸收后,产生10.0g白色沉淀,则a的数值为( )
| A. | 5.4g | B. | 6.2g | C. | 7.8g | D. | 9.0g |
18.下列四个有关燃烧的实验解释错误的是( )
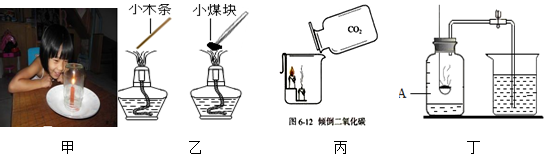

| A. | 甲图实验中,瓶内的蜡烛燃烧一会儿就熄灭了,说明燃烧需要氧气 | |
| B. | 乙图中,小木条马上被点燃,小煤块点不燃,是因为煤块的着火点比较高 | |
| C. | 丙图实验中,蜡烛火焰自下而上熄灭,说明CO2密度比空气大 | |
| D. | 丁图测量空气中的氧气含量实验,把燃烧匙内红磷换成木炭效果会更好 |
15.下列实验设计,不能达到实验目的是( )
| A. | 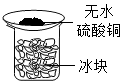 装置验证空气中有水分 | B. | 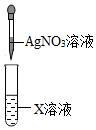 装置验证X溶液中是否含有Cl- | ||
| C. | 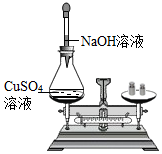 装置验证质量守恒定律 | D. | 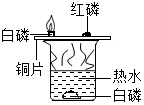 装置验证可燃物燃烧的两个条件 |
12.某市发生了一起亚硝酸钠中毒事件.亚硝酸钠外观酷似食盐,有咸味,加热会分解,放出有臭味的气体.请结合下表信息,判断以下区别亚硝酸钠和氯化钠的方法错误的是( )
| 亚硝酸钠(NaNO2) | 氯化钠(NaCl) | |
| 跟稀盐酸作用 | 放出红棕色的气体 | 无反应 |
| 跟硝酸银溶液作用 | 生成浅黄色沉淀 | 生成白色沉淀 |
| A. | 滴加稀盐酸 | B. | 加热 | C. | 品尝味道 | D. | 滴加硝酸银溶液 |
19.现有一组物质:海水、盐酸、酒精溶液,下列物质中,可以和这组物质归为同一类的是( )
| A. | 冰水 | B. | 泥水 | C. | 氨水 | D. | 原油 |