��Ŀ����
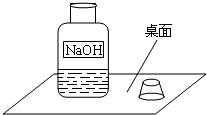
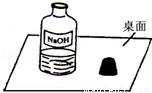
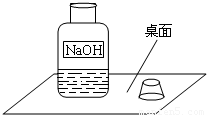
һ�죬С���߽�ʵ���ң�������һ��������г���Ļ��棨����ͼ����
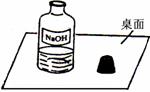 Χ�ƴ�ƿ�Լ��Ƿ���ʵ����⣬չ����̽����
Χ�ƴ�ƿ�Լ��Ƿ���ʵ����⣬չ����̽����
��1��С��������в��룺
�� ��һ������Լ���ȫ���ʣ�����Һ�������� ��
��һ������Լ���ȫ���ʣ�����Һ�������� ��
�����������Լ����ֱ��ʣ�����Һ��������NaOH��Na2CO3��
������������Լ�û�б��ʣ�����Һ��������NaOH��
��2��д��NaOH�������ʵĻ�ѧ��Ӧ����ʽ��
��
��3���������ʵ��֤ʵ���Լ��Ѿ����ֱ��ʣ����̽��������
| ʵ�鲽�� | Ԥ�������� |
|
|
��1��Na2CO3(��̼����)��1�֣���
��2��2NaOH+ CO2 == Na2CO3 + H2O��2�֣�
��3����6�֣�
| ʵ����� | ʵ�������� |
| ȡ��������Һ���Թܣ���������BaCl2��Һ��1�֣� | ������ɫ������1�֣�����˵����Һ�к���Na2CO3��1�֣� |
| ���ú�������Թ��У��μ�������̪��Һ��1�֣� | ��Һ��죨1�֣�����˵����Һ�к���NaOH��1�֣� |
����ע��������������ʵ�ʵ�鷽����ȷ����3�֡����У�û��ȡ����1�֣�BaCl2��Һû��д����������1�֣���������1�֣����۴����1�֣������ָ��֡����ʵ�鷽�������֡���
��������ѡ�Լ��������ɣ�

 ӥ�ɽ̸��νӽ̲ĺӱ�����������ϵ�д�
ӥ�ɽ̸��νӽ̲ĺӱ�����������ϵ�д� ���������ν�ϵ�д�
���������ν�ϵ�д�