��Ŀ����
18�� ������һ���Ż��ֻ��40oC�Ŀ�ȼ������������ð����еĿ���������������ʵ��̽����
������һ���Ż��ֻ��40oC�Ŀ�ȼ������������ð����еĿ���������������ʵ��̽��������ƿ����װ������a�������ˮ���ھƾ��Ƶ����ϰѲ������¶˼��ȣ�װ����ͼ�ܷ⣮�����Ѳ�������������Ӵ�������ȼ�գ���ʼʱƿ������ˮѹ���ձ��У��Ժ��ձ���ˮ��ѹ��ƿ�У�ȼ��ֹͣ��������ȫ��ȴ����������ƿ��ˮ�������b������������ƿ�����c�����ش��������⣺
��1���¶��ȵIJ������ڸ�ʵ�����������������ȼ���ף�
��2������ȼ��ʱ��ƿ�в����������ų��������÷�Ӧ�ķ��ű���ʽΪ4P+5O2$\frac{\underline{\;��ȼ\;}}{\;}$2P2O5��
��3���á�a��b��c���Ĵ���ʽ�ش𣬸�ʵ����ƿ�����������Ϊb-a������������������Ϊ��b-a������c-a����
��4����ʵ���м���ƿ����װ������a�������ˮ������Ҫ�����Ǽӿ켯��ƿ����ȴ�����գ����ܽ⣩����ȼ�����ɵ����������ף�
���� ��1�������¶��ȵIJ������ڸ�ʵ�����������������ȼ�����н��
��2�����ݰ���ȼ��ʱ��ƿ�в����������̣��ų��������н��
��3�����ݽ���ˮ���������������������н��
��4������ˮ�ܽ����Լ���������������Ӧ�������
��� �⣺��1���¶��ȵIJ������ڸ�ʵ�����������������ȼ���ף�
��2������ȼ��ʱ��ƿ�в����������̣��ų��������������ڵ�ȼ���������������������ף��÷�Ӧ�Ļ�ѧ����ʽΪ��4P+5O2$\frac{\underline{\;��ȼ\;}}{\;}$2P2O5��
��3������ˮ�����������������������Ը�ʵ����ƿ�����������Ϊb-a������������������Ϊ��b-a������c-a����
��4�������¶ȵĸı��Ӱ��ˮ�Ľ��룬������Ҫʹ����ƿ�¶Ⱦ��콵�������£�ͬʱˮҲ�����ղ������ɵ�������������һ�����ã�
�ʴ�Ϊ����1����ȼ���ף�
��2�����̣�4P+5O2$\frac{\underline{\;��ȼ\;}}{\;}$2P2O5��
��3��b-a����b-a������c-a����
��4���ӿ켯��ƿ����ȴ�����գ����ܽ⣩����ȼ�����ɵ����������ף�
���� ���������ð��ײⶨ�����ڿ����е��������������ʵ�鷽�����̵�̽��������̽��ʵ��Ҫ����������������������ƵIJ����в�ͬ������ȥ˼����ȥ̽�����Ӷ��ش���Ŀ���������⣮��������Ҫ������������ʵ�����У�

 ����С��ʿ���������ϵ�д�
����С��ʿ���������ϵ�д�| A�� | Һ�� | B�� | ʯ��ˮ | C�� | ��Ȫˮ | D�� | ���� |
| A�� | �˵����⡢ƻ������ | B�� | ���;��ӷ�����ֲ��ĺ������� | ||
| C�� | �۱��۶ϡ�ֽ��ȼ�� | D�� | ֲ�������á�ѩ���ڻ� |

| A�� | þԪ�ص�ԭ������Ϊ12 | |
| B�� | ��ʾ�����ӵĽṹʾ��ͼ��E | |
| C�� | þԪ������Ԫ�ص���ʵ��������������������� | |
| D�� | �ȵ����ԭ������Ϊ35.45 |
 �����ڿ�����ȼ�գ�����ֻ��������Ӧ����Ҳ�����⣬��þ�ڿ�����ȼ�ղ�������������Ӧ�������뵪���Ͷ�����̼��Ӧ����ͼ�Dzⶨ����������������ʵ��װ��ͼ��ȼ�ճ��ڷ�����ף��������Լռ�����������$\frac{1}{5}$��
�����ڿ�����ȼ�գ�����ֻ��������Ӧ����Ҳ�����⣬��þ�ڿ�����ȼ�ղ�������������Ӧ�������뵪���Ͷ�����̼��Ӧ����ͼ�Dzⶨ����������������ʵ��װ��ͼ��ȼ�ճ��ڷ�����ף��������Լռ�����������$\frac{1}{5}$��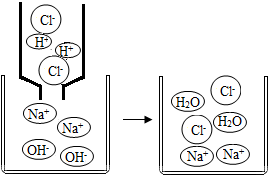 ��ͼ�����л�ѧ�У�����ѧϰ����ͼ���кͷ�Ӧ��ʵ����H++OH-=H2O��������ʵ�ʲμӷ�Ӧ�����ӷ�������ʾ��Ӧ��ʽ�ӽ����ӷ���ʽ�����ӷ���ʽ����дһ�㰴���²��裺����Na2SO4��BaCl2��ӦΪ����
��ͼ�����л�ѧ�У�����ѧϰ����ͼ���кͷ�Ӧ��ʵ����H++OH-=H2O��������ʵ�ʲμӷ�Ӧ�����ӷ�������ʾ��Ӧ��ʽ�ӽ����ӷ���ʽ�����ӷ���ʽ����дһ�㰴���²��裺����Na2SO4��BaCl2��ӦΪ����