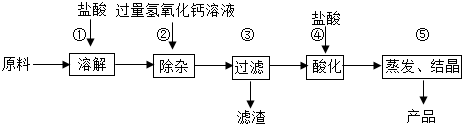
��Ŀ����
6����ͨ��״���£�������һ�ֻ���ɫ���壬�ܶȱȿ�����������ˮ���ڹ�ҵ�ϣ���ȡ��������Ҫ����������ͼ��
��1�����÷紵��ɹ���ԴӺ�ˮ����ȡ���Σ��ù�����Ҫ�������� ������������ѧ�����仯��
��2��������������ʹ�������Ȼ�����Һ��Ϊ������Һ�ķ����ֱ�������ˮ�ֺ��������ʣ�
��3��Ҫ��ȥʳ��ˮ�л��е�������ɳ����ѡ��IJ��������ǹ��ˣ�
��4��д������Ȼ��Ʊ�����Һ��ȡ�����Ļ�ѧ����ʽ2NaCl+2H2O$\frac{\underline{\;ͨ��\;}}{\;}$2NaOH+H2��+Cl2����
���� ��1���������÷紵��ɹ�Ӻ�ˮ����ȡ���εĹ����У�ˮ�����������Ȼ��Ƶ���������Խ��Խ��û�����������ɽ��з�����
��2�����ݱ�����Һ�Ķ�����з�����
��3�����ݹ��˿��Գ�ȥ������ˮ�����ʽ��з�����
��4���������еķ�Ӧԭ���ҳ���Ӧ������P��Ӧ������Ȼ�����������غ㶨����д��ѧ����ʽ��
��� �⣺��1�����÷紵��ɹ�Ӻ�ˮ����ȡ���εĹ����У�ˮ�����������Ȼ��Ƶ���������Խ��Խ��û�����������ɣ����Ըù�����Ҫ���������仯��
��2��������Һʱ�������ܽ�ij�����ʵ���Һ���������������IJ������Ȼ�����Һ�У���������Һ��û�дﵽ�����ܽ��������Ա�Ϊ������Һ�ķ����ֱ��ǣ�����ˮ�֣��������ʣ�
��3��Ҫ��ȥʳ��ˮ�л��е�������ɳ����ѡ��IJ��������ǣ����ˣ�
��4���Ȼ�����Һ��ͨ��������������������ơ���������������ѧ����ʽΪ��2NaCl+2H2O$\frac{\underline{\;ͨ��\;}}{\;}$2NaOH+H2��+Cl2����
�ʴ�Ϊ����1��������
��2������ˮ�֣��������ʣ�
��3�����ˣ�
��4��2NaCl+2H2O$\frac{\underline{\;ͨ��\;}}{\;}$2NaOH+H2��+Cl2����
���� �����Ҫ���ջ�ѧ����ʽ����д�������˵ȷ�������ݣ�ֻ���������ܶ���ط��������������ȷ���жϣ�

 ��У����ϵ�д�
��У����ϵ�д����¶ȡ����������ʿ�����С�������������������������ܼ��������������ܼ����࣮
| A�� | �٢� | B�� | �٢� | C�� | �ڢۢ� | D�� | �٢ܢ� |
| A�� |  ���������� | B�� |  Ϩ��ƾ��� | C�� |  ��ȡҺ�� | D�� |  ���������� |
| A�� | ��һ�ȶ���Һ�������Һ | B�� | ���ͳ��������黯���� | ||
| C�� | ѩ��������Ϊ��������̵� | D�� | Ũ��Һ��ˮϡ�ͺ������������� |
| A�� | �÷���ˮ����Ӳˮ����ˮ | B�� | ��ʳ��ˮ������ | ||
| C�� | ��ʳ�ô׳�ȥˮ�����ˮ�� | D�� | ��ˮ������ʯ�Һ���ʯ�� |
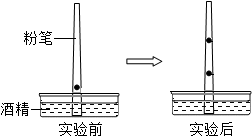 �����ھ�һֻ�۱ʵĴֶ�2cm������һ����īˮ�����۱ʲ���ʢ�оƾ���Һ���������У�����һ��ʱ�䣬���������žƾ�����������īˮҲ�ڷ۱��ϲ����ƶ��������īˮ�ڷ۱��Ϸ�Ϊ���������ϰ벿������ɫ���°벿������ɫ����ͼ��ʾ����������������ش��������⣺
�����ھ�һֻ�۱ʵĴֶ�2cm������һ����īˮ�����۱ʲ���ʢ�оƾ���Һ���������У�����һ��ʱ�䣬���������žƾ�����������īˮҲ�ڷ۱��ϲ����ƶ��������īˮ�ڷ۱��Ϸ�Ϊ���������ϰ벿������ɫ���°벿������ɫ����ͼ��ʾ����������������ش��������⣺