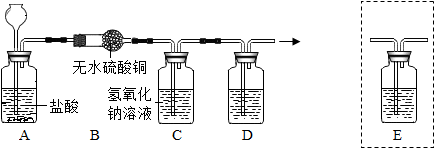
��Ŀ����
16����ȷ�IJ��������ܱ�֤��ѧʵ���˳�����У����ܷ�ֹ�����¹ʵķ��������ж��������¹ʵ�Ԥ��������ȷ���ǣ�������| ѡ�� | �����¹� | Ԥ���������� |
| A | ���������ϵľƾ�ȼ������ | ������ʪĨ����ɳ�Ӹ��� |
| B | Ũ���������Ƥ���� | ���ô���ˮ��ϴ����Ϳ��3%��5%��С�մ���Һ |
| C | ��ʴ��ҩҺ�����۾� | ���������۾�����������ˮ��ϴ |
| D | ��Һ���������·��� | ����ˮ��ϴ����Ϳ��������Һ |
| A�� | A | B�� | B | C�� | C | D�� | D |
���� A�����ݾƾ�ȼ��������������ʪĨ����ɳ�����𣬽��º��������������
B������С�մ���Һ�ʼ��ԣ���Ӧ��������Ƥ���ϵ���Һ�������˺����
C������ҩҺ�����۾��¹ʴ����������н��
D�������кͼ�Һʱ������ǿ����Һ���н��
��� �⣺A�����������ϵľƾ�ȼ��������������ʪĨ����ɳ������������ȷ����Ϊ���º�����������𣬼�ʱ��Ч������ȷ��
B��Ũ����к�ǿ�ĸ�ʴ�ԣ���������Ƥ���ϣ�������Ũ������õ���Ȼ���ô���ˮ��ϴ��ϴ����Ϳ��3%��5%��С�մ���Һ���ʴ���
C��ʵ��ҩҺ�����۾���������ˮ��ϴ���в���������꣬�ʴ���
D���кͼ�Һ������ǿ�ᣬ������ɶ��θ�ʴ���ʴ���
��ѡ��A��
���� ����һЩʵ�����ע�������̽�����˽ⳣ���������¹ʵĴ��������ͼ��ɣ��ѶȲ���

��ϰ��ϵ�д�
�����Ŀ
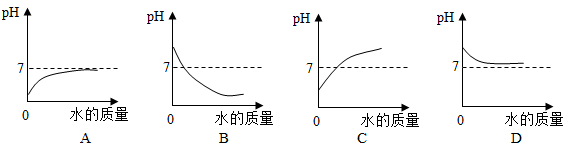
4���������ʵ���Һ���ڷ����ڿ����У���Һ����������ѧ�仯����С���ǣ�������
| A�� | Ũ���� | B�� | ����������Һ | C�� | Ũ���� | D�� | ����������Һ |
11������С���ͬѧ�ڲⶨ��NaCl��Na2CO3�γɵĹ����������ʱ������������ʵ�飺ȡ40g�������������Һ��ƽ����Ϊ�ķݣ�Ȼ��ֱ����һ������������CaCl2��Һ��ʵ�����ݼ��±���
������������ݻش��㣺
��1�����ɵij����ǣ���д��ѧʽ��CaCO3��
��2��10gԭ����������ɵ���Һ������CaCl2��Һ��Ӧ��������ɳ�������Ϊ5g��
��3��m=4g��
��4��ԭ����������NaCl�����������Ƕ��٣�
������������ݻش��㣺
| ʵ��һ | ʵ��� | ʵ���� | ʵ���� | |
| ԭ������������ | 10g | 10g | 10g | 10g |
| ����CaCl2��Һ���� | 10g | 20g | 30g | 40g |
| ���ɵij��������� | 2g | m | 5g | 5g |
��2��10gԭ����������ɵ���Һ������CaCl2��Һ��Ӧ��������ɳ�������Ϊ5g��
��3��m=4g��
��4��ԭ����������NaCl�����������Ƕ��٣�
1����ѧ��Դ�������ַ��������
�±��������г���������Һ��pH����Ҫ�ɷֻ�ѧʽ��
�����ϱ���Ϣ�ش��������⣺
���������У�������ǿ���Ǣܣ�����ţ��������Ʒ䶣ҧ���ͷ�һ�ּ������ʣ���Ϊ����֢״�����ڶ�ҧ��Ϳ�٣�����ţ�
�±��������г���������Һ��pH����Ҫ�ɷֻ�ѧʽ��
| ��� | �� | �� | �� | �� | �� |
| �� �� | ʳ �� | �� �� | ����ˮ | ʯ��ˮ | ������Һ |
| ��Ҫ�ɷ� | CH3COOH | C2H5OH | C12H22O11 | Ca��OH��2 | Na2CO3 |
| ��ҺpH | 3 | 7 | 7 | 11 | 8 |
���������У�������ǿ���Ǣܣ�����ţ��������Ʒ䶣ҧ���ͷ�һ�ּ������ʣ���Ϊ����֢״�����ڶ�ҧ��Ϳ�٣�����ţ�
8��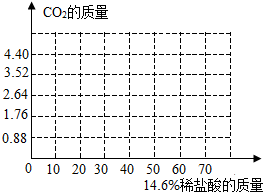 С��ͬѧΪ�˲ⶨ������Ʒ��̼�������Ȼ��ƵĻ�����̼���Ƶ�����������ȡ��Ʒ13.9g�����ձ��У���60g��������Ϊ14.6%��ϡ�����6�μ��뵽����Ʒ�У���ַ�Ӧ����ѧ����ʽΪ��Na2CO3+2HCl�T2NaCl+CO2��+H2O����÷�Ӧ���������������m���������±���ʾ��
С��ͬѧΪ�˲ⶨ������Ʒ��̼�������Ȼ��ƵĻ�����̼���Ƶ�����������ȡ��Ʒ13.9g�����ձ��У���60g��������Ϊ14.6%��ϡ�����6�μ��뵽����Ʒ�У���ַ�Ӧ����ѧ����ʽΪ��Na2CO3+2HCl�T2NaCl+CO2��+H2O����÷�Ӧ���������������m���������±���ʾ��
������Ŀ�ش��������⣺
��1��������Ʒ��ȫ��Ӧ������CO2������Ϊ4.4g��
��2��������ͼ����ͼ�л�����Ӧ���������������m����ϡ���������仯�����ߣ�
��3����Ʒ��̼���Ƶ���������Ϊ76.3%��������������0.1%������д��������̣�
 С��ͬѧΪ�˲ⶨ������Ʒ��̼�������Ȼ��ƵĻ�����̼���Ƶ�����������ȡ��Ʒ13.9g�����ձ��У���60g��������Ϊ14.6%��ϡ�����6�μ��뵽����Ʒ�У���ַ�Ӧ����ѧ����ʽΪ��Na2CO3+2HCl�T2NaCl+CO2��+H2O����÷�Ӧ���������������m���������±���ʾ��
С��ͬѧΪ�˲ⶨ������Ʒ��̼�������Ȼ��ƵĻ�����̼���Ƶ�����������ȡ��Ʒ13.9g�����ձ��У���60g��������Ϊ14.6%��ϡ�����6�μ��뵽����Ʒ�У���ַ�Ӧ����ѧ����ʽΪ��Na2CO3+2HCl�T2NaCl+CO2��+H2O����÷�Ӧ���������������m���������±���ʾ��| ʵ����� | ��1�� | ��2�� | ��3�� | ��4�� | ��5�� | ��6�� |
| ����ϡ����������g�� | 10 | 10 | 10 | 10 | 10 | 10 |
| ����������������g�� | 0.88 | 1.76 | 2.64 | 3.52 | 4.4 | 4.4 |
��1��������Ʒ��ȫ��Ӧ������CO2������Ϊ4.4g��
��2��������ͼ����ͼ�л�����Ӧ���������������m����ϡ���������仯�����ߣ�
��3����Ʒ��̼���Ƶ���������Ϊ76.3%��������������0.1%������д��������̣�
5�����в����ڻ����������ǣ�������
| A�� | ��������ȼ�� | B�� | ʳ��ĸ��� | C�� | ������ | D�� | �������� |