��Ŀ����
Ϊ�ⶨCuһZn�Ͻ����ɣ�С��ͬѧ���øúϽ��ĩ��ϡ���ᷴӦ������������ʵ�飬�������ʵ�����ݼ�¼���£�
| ��һ�� | �ڶ��� | ������ | |
| ��ȡ�Ͻ������/g | 1O | 10 | 20 |
| �������������/g | 50 | 80 | 50 |
| ��������������/g | 0.2 | 0.2 | 0.2 |
����㣺
(1)���ϱ����ݷ���������ȡ�Ͻ�������ϡ�����������Ϊ ʱ�������Ͻ��е�п��ϡ����ǡ����ȫ��Ӧ��
(2)�úϽ���п������Ϊ���ٿ�?(����������һλС��)
(3)���úϽ��е�п��ϡ����ǡ����ȫ��Ӧʱ������Һ�����ʵ���������Ϊ����?(���� �����ȷ��0.1��)
��1��1��5 (1��)
��2���⣺��Ͻ���п������x,Ϊ��������п������Ϊy
Zn + H2SO4 ==== ZnSO4 + H2�� (1��)
65 161 2
x y 0.2 g
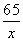 =
= 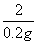 x = 6.5 g (1��)
x = 6.5 g (1��)
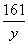 =
=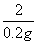 y = 16.1 g (1��)
y = 16.1 g (1��)
��3��������Һ������п������������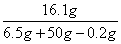 �� 100% = 28.6% (2��)
�� 100% = 28.6% (2��)
�𣺣��ԣ�

��ϰ��ϵ�д�
�����Ŀ
 ��������ʯ��
��������ʯ��  ��
��