��Ŀ����
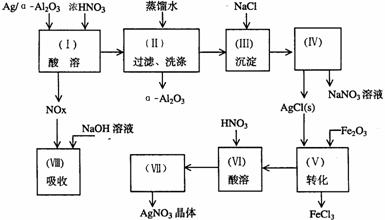 Ag/�����ͣ�Al2O3��ʯ�ͻ�ѧ��ҵ��һ����Ҫ����������Ag������ã�(����)Al2O3�������Ҳ��������ᡣ�ô����Ļ���ʵ������ͼ��ʾ��
Ag/�����ͣ�Al2O3��ʯ�ͻ�ѧ��ҵ��һ����Ҫ����������Ag������ã�(����)Al2O3�������Ҳ��������ᡣ�ô����Ļ���ʵ������ͼ��ʾ��

���У�������ת����ӦΪ��6AgCl��Fe2O3 = 3Ag2O��2FeCl3
 �����������շ�ӦΪ��2NaOH+NO2+NO=2NaNO2+H2O
�����������շ�ӦΪ��2NaOH+NO2+NO=2NaNO2+H2O
���Ķ�����ʵ�����̣����������գ�
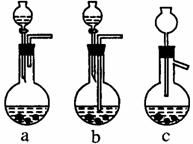
��1���ڣ�����Ag/(����)Al2O3�����ܽ�Ӧ��ѡ����ͼװ���е� ��ѡ��a��b��c����
�˴���Ӧ�Ļ�ѧ����ʽΪ��
��
��2���ڣ����������������ˮ��������ˮ����ϴ�ӣ����ᷢ����ѧ��Ӧ�����ӷ���ʽΪ�� ��
��3����(��)����Ӧ�Ļ�ѧ����ʽΪ�� ��
��4����֪��ʵ������AgNO3��Һ��Ҫ����ɫƿ�ܹⱣ�档�ڵڣ��������д�AgNO3��Һ���AgNO3������Ҫ���е�ʵ���������Ϊ ������ţ���
A. ���� B. ���� C. ���� D. ���� E. ��ȴ�ᾧ F.��ȡ
��1��a��1�֣���Ag+2HNO3��Ũ��=AgNO3+NO2��+H2O��3Ag+4HNO3(ϡ)=3AgNO3+NO��+2H2O����3�֣���6�֣�
��2��Ag++Cl-=AgCl��(2��)
��3��Ag2O+2HNO3=2AgNO3+H2O(3��)
��4��B��E��D��2�֣�

�����ڽ���Ԫ�ؼס�����Ԫ�����ڱ������λ�������ʾ�������ж���ȷ����
| �� | �� | |
| �� | �� | �� |
A������������ԣ�����������
B�������ԣ��ף���
C��ԭ�Ӱ뾶������������
D���������������ף���
���ⶨNa2SO4�ڲ�ͬ�¶�ʱ���ܽ���������±���
| �¶�/�� | 40 | 60 | 80 | 100 |
| �ܽ��/ g | 48��8 | 45.3 | 43.7 | 42.5 |
��ͼ�DZ�ʾ���¶ȱ仯������Һ�������������ı仯���ߣ������ܹ���ʾ�����¶ȱ仯������Һ���������������仯���ߵ���( )
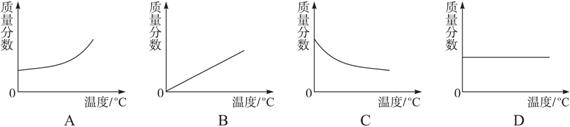
 ��Һ�����������仯���ɵ���
��Һ�����������仯���ɵ���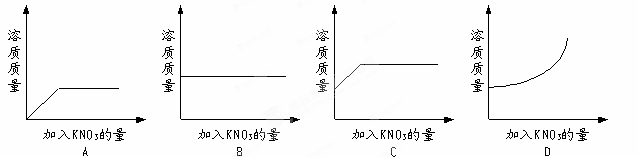
 .̼泥�NH4 HCO3��
.̼泥�NH4 HCO3��