��Ŀ����
ij��ѧ��ȤС���ͬѧ��ʵ����������������Ϊ20%��̼������Һ��������й�ʵ�飮
��1������100g��������Ϊ20%��̼������Һ��
�����Ʒ���A����Ҫ̼���ƹ��������Ϊ g��ˮ�����Ϊ mL��ˮ���ܶȽ��ƿ���1g/cm3����
���Ʒ���B������40%��̼������Һϡ�ͳ�100��20%̼������Һ������ȡ40%��̼������Һ�������������ܶ�Ϊ1.4��/������ ��
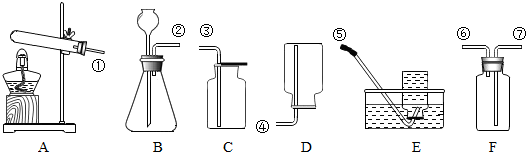
������������������һ����̼������Һ�������²���˳����У�
������ͼ��ʾ����ű�ʾ��ȷ���Ƹ���Һ�IJ���˳��Ϊ ��
����������ƽ���������̼����ʱ������������ƽ��ָ��ƫ�����̣�Ӧ ��
A�������������� B��������������
C���������� D����������
�ܷ���������ԭ����ܵ����䵽��̼������Һ������������С��8%����
A�������л������� B����ˮʱ���Ӷ���
C����ú�װ���Լ�ƿ��ʱ������Һ������ D����ˮʱ���Ӷ���
��2��ȡ�������Ƶõ���20%��̼������Һ53�ˣ�����68.4��ijŨ�ȵ�ϡ�����ǡ����ȫ��Ӧ����Ӧ��������Һ����������������
��1������100g��������Ϊ20%��̼������Һ��
�����Ʒ���A����Ҫ̼���ƹ��������Ϊ
���Ʒ���B������40%��̼������Һϡ�ͳ�100��20%̼������Һ������ȡ40%��̼������Һ�������������ܶ�Ϊ1.4��/������
������������������һ����̼������Һ�������²���˳����У�

������ͼ��ʾ����ű�ʾ��ȷ���Ƹ���Һ�IJ���˳��Ϊ
����������ƽ���������̼����ʱ������������ƽ��ָ��ƫ�����̣�Ӧ
A�������������� B��������������
C���������� D����������
�ܷ���������ԭ����ܵ����䵽��̼������Һ������������С��8%����
A�������л������� B����ˮʱ���Ӷ���
C����ú�װ���Լ�ƿ��ʱ������Һ������ D����ˮʱ���Ӷ���
��2��ȡ�������Ƶõ���20%��̼������Һ53�ˣ�����68.4��ijŨ�ȵ�ϡ�����ǡ����ȫ��Ӧ����Ӧ��������Һ����������������
���㣺һ������������������Һ������,������-������ƽ,�й��������������ļ���,���ݻ�ѧ��Ӧ����ʽ�ļ���
ר�⣺��Һ����Һ���ܽ��
��������1���ٸ������ʵ�����=��Һ�����������ʵ�����������ˮ������=��Һ������-���ʵ����������=
���Լ���Һϡ��������������������н��
�ڸ���������Һ��ʵ�鲽����н��
�۸�����������ƽ���������̼����ʱ������������ƽ��ָ��ƫ�����̣�˵��ҩƷ����н��
�ܸ�����Һ����ʱ̼������Һ������������С��8%�����������ʼ��ٻ��ܼ�������н��
��2������̼���Ƶ�����������ɶ�����̼�������Լ��Ȼ��Ƶ����������������Ӧ��������Һ����������������
| ���� |
| �ܶ� |
�ڸ���������Һ��ʵ�鲽����н��
�۸�����������ƽ���������̼����ʱ������������ƽ��ָ��ƫ�����̣�˵��ҩƷ����н��
�ܸ�����Һ����ʱ̼������Һ������������С��8%�����������ʼ��ٻ��ܼ�������н��
��2������̼���Ƶ�����������ɶ�����̼�������Լ��Ȼ��Ƶ����������������Ӧ��������Һ����������������
����⣺��1������Ҫ̼���ƹ��������=100g��20%=20g��ˮ������100g-20g=80g��ˮ�����=
=80mL��
��40%��̼������Һ�����Ϊx
100g��20%=40%��x��1.4g/mL
x=35.7mL
�ʴ�Ϊ��20��80��35.7mL��
�����Ƹ���Һ�IJ���˳��Ϊ���㡢��������ȡ���ܽ⣬�������ΪCBDEA���ʴ�Ϊ��CBDEA��
����������ƽ���������̼����ʱ������������ƽ��ָ��ƫ�����̣�˵��ҩƷ�࣬����Ӧ���Ǽ����������壻�ʴ�Ϊ��B��
��A�������л������ʣ���ʹ������������ƫС��B����ˮʱ���Ӷ����ᵼ���ܼ����࣬��ʹ������������ƫС��
C����ú�װ���Լ�ƿ��ʱ������Һ����������Һ����ʧ�����������������䣻D����ˮʱ���Ӷ����ᵼ���ܼ����٣���ʹ������������ƫ��
�ʴ�Ϊ��AB��
��2�������ɶ�����̼������Ϊx�������Ȼ��Ƶ�����Ϊy��
Na2CO3+2HCl=2NaCl+H2O+CO2��
106 117 44
20%��53g y x
=
x=4.4g
=
y=11.7g
��Ӧ��������Һ��������������=
��100%=10%
�𣺷�Ӧ��������Һ��������������Ϊ10%��
| 80g |
| 1g/mL |
��40%��̼������Һ�����Ϊx
100g��20%=40%��x��1.4g/mL
x=35.7mL
�ʴ�Ϊ��20��80��35.7mL��
�����Ƹ���Һ�IJ���˳��Ϊ���㡢��������ȡ���ܽ⣬�������ΪCBDEA���ʴ�Ϊ��CBDEA��
����������ƽ���������̼����ʱ������������ƽ��ָ��ƫ�����̣�˵��ҩƷ�࣬����Ӧ���Ǽ����������壻�ʴ�Ϊ��B��
��A�������л������ʣ���ʹ������������ƫС��B����ˮʱ���Ӷ����ᵼ���ܼ����࣬��ʹ������������ƫС��
C����ú�װ���Լ�ƿ��ʱ������Һ����������Һ����ʧ�����������������䣻D����ˮʱ���Ӷ����ᵼ���ܼ����٣���ʹ������������ƫ��
�ʴ�Ϊ��AB��
��2�������ɶ�����̼������Ϊx�������Ȼ��Ƶ�����Ϊy��
Na2CO3+2HCl=2NaCl+H2O+CO2��
106 117 44
20%��53g y x
| 106 |
| 20%��53g |
| 44 |
| x |
x=4.4g
| 106 |
| 20%��53g |
| 117 |
| y |
y=11.7g
��Ӧ��������Һ��������������=
| 11.7g |
| 53g+68.4g-4.4g |
�𣺷�Ӧ��������Һ��������������Ϊ10%��
�����������ܽϺÿ���ѧ����˼ά�������ͷ������������������������֪ʶ������ȷ�����ͽ��

��ϰ��ϵ�д�
�����Ŀ
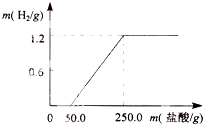 ij��ȤС��ӷ������ײ���һ����Ƭ����������21.9%��ϡ�����У������������������������������������ͼ��������������Ĥ��Ӧʱû��H2�������������ʲ����ᷴӦ������ش�
ij��ȤС��ӷ������ײ���һ����Ƭ����������21.9%��ϡ�����У������������������������������������ͼ��������������Ĥ��Ӧʱû��H2�������������ʲ����ᷴӦ������ش�