��Ŀ����
������ͼ�ĸ���ѧʵ�飬�ش����⣺����ʾ��Cu��Zn�ĺϽ��Ϊ��ͭ��
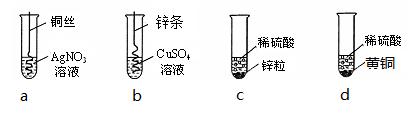
������ʵ��a�е�ʵ������ (12) ��
��Ҫ֤��Cu��Zn��Ag�Ļ��˳����Ҫ����ʵ���� ��13�� ����ʵ����ţ���
����cʵ����ʣ����Һ�У�һ���е������� ��14�� �������е��� ���� ��15�� ��Ϊ��ȷ�������е����ʣ��ɼ��� ��16�� ����ѡ����ţ�
���� ��15�� ��Ϊ��ȷ�������е����ʣ��ɼ��� ��16�� ����ѡ����ţ�
���Ȼ�����Һ ��������ͭ ������������Һ ����п��
��Ϊ�˲ⶨd��ϡ�������������������ȡ20g�û�ͭ��Ʒ���ձ��У������з�4�μ����ϡ���ᣬ��ַ�Ӧ��ÿ������ϡ�����������ʣ������������¼���±���
| ����ϡ�����������g�� | ��ַ�Ӧ��ʣ������������g�� | |
| ��1�� | 25 | 16.75 |
| ��2�� | 25 | 13.5 |
| ��3�� | 25 | m |
| ��4�� | 2 | 12.0 |
�Իش��������⣺
������������m��ֵΪ ��17�� ��
��ϡ������������������Ƕ��٣������ݻ�ѧ����ʽ���㣩
(12) ͭƬ����������ɫ�������� (13) a b
(14) ZnSO4 (15) H2 SO4 (16) �� �� (17) 12
��H2 SO4 x mol
20-16.75/65=0.05 ---------1��
Zn + H2 SO4 �� ZnSO4+H2�� -------1��
1 1
0.05 mol x mol
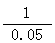 =
=  x = 0.05 mol -----1�� 0.05��98/25=0.196����19.
x = 0.05 mol -----1�� 0.05��98/25=0.196����19. 6%��---1��
6%��---1��

 �����Ծ���ĩ���100��ϵ�д�
�����Ծ���ĩ���100��ϵ�д� ˫��ͬ������ѵ��ϵ�д�
˫��ͬ������ѵ��ϵ�д� Si+2CO����ȡ�裬�÷�Ӧ��
Si+2CO����ȡ�裬�÷�Ӧ��