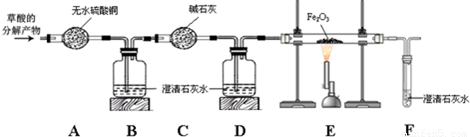
��Ŀ����
��ͼ�Ƕ�����̼����֪ʶ����������ͼ�����ַ�Ӧ�����Ͳ���������ʡ�ԣ����밴����Ҫ����գ�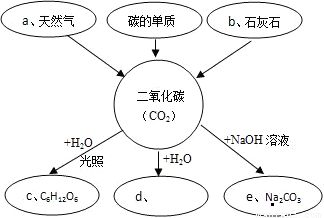
��1������a��Ҫ�ɷֵĻ�ѧʽ��______��
��2������b��Ҫ�ɷ�����������______��
��3������c��C6H12O6�����������______���������л��������������C��H��O����Ԫ�ص���������______��
��4��д����CO2��������d�Ļ�ѧ��Ӧ����ʽ______��
��5���������e��ѧʽ�д���Ԫ�صĻ��ϼ�______��
���𰸡�����������ͼʾ������֮��ķ�Ӧ��ϵȷ��δ֪���ʵĻ�ѧʽ���ٽ����������еڣ�3���ʣ����ݻ�ѧʽ���Ԫ�ص������Ⱦ��Ǹ�Ԫ�ص����ԭ����������ԭ�Ӹ�����Ȼ��������ý�����ڣ�5���ʿɸ��ݳ�����̼���Ϊ-2�ۣ����ݻ������л��ϼ۴�����Ϊ���ԭ����н��
����⣺����Ȼ������Ҫ�ɷ��Ǽ��飬��ѧʽ��CH4��
��ʯ��ʯ����Ҫ�ɷ���̼��ƣ�CaCO3�������ɸ����Ӻ�̼������ӹ��ɵĻ�������е��������Ǹ����ӣ�Ca2+����
������c��C6H12O6����̼Ԫ�������л��
����C��H��O����Ԫ�ص��������ǣ�Ar��C��×6��Ar��H��×12��Ar��O��×6=12×6��1×12��16×6=6��1��8��
�ܶ�����̼��ˮ��Ӧ����̼�ᣬ��ѧ����ʽΪ��CO2+H2O=H2CO3
������e�Ļ�ѧʽNa2CO3������̼�������-2�ۣ����ݻ������и�Ԫ�صĻ��ϼ۴�����Ϊ�㣬��֪��Ԫ�صĻ��ϼ�Ϊ+1�ۣ�
�ʴ�Ϊ��
��1��CH4��
��2��Ca2+��
��3����6��1��8��
��4��CO2+H2O=H2CO3 ��
��5��+1�ۣ�
������̼���仯����֮���ת���Ŀ������п��г�����֪ʶ��ϵ��ѧ��ƽʱҪ��������ʵ����ʽ����������ã�
����⣺����Ȼ������Ҫ�ɷ��Ǽ��飬��ѧʽ��CH4��
��ʯ��ʯ����Ҫ�ɷ���̼��ƣ�CaCO3�������ɸ����Ӻ�̼������ӹ��ɵĻ�������е��������Ǹ����ӣ�Ca2+����
������c��C6H12O6����̼Ԫ�������л��
����C��H��O����Ԫ�ص��������ǣ�Ar��C��×6��Ar��H��×12��Ar��O��×6=12×6��1×12��16×6=6��1��8��
�ܶ�����̼��ˮ��Ӧ����̼�ᣬ��ѧ����ʽΪ��CO2+H2O=H2CO3
������e�Ļ�ѧʽNa2CO3������̼�������-2�ۣ����ݻ������и�Ԫ�صĻ��ϼ۴�����Ϊ�㣬��֪��Ԫ�صĻ��ϼ�Ϊ+1�ۣ�
�ʴ�Ϊ��
��1��CH4��
��2��Ca2+��
��3����6��1��8��
��4��CO2+H2O=H2CO3 ��
��5��+1�ۣ�
������̼���仯����֮���ת���Ŀ������п��г�����֪ʶ��ϵ��ѧ��ƽʱҪ��������ʵ����ʽ����������ã�

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ




 ��д��ͼ�д�������������ƣ��� ��
��д��ͼ�д�������������ƣ��� ��